Address
304 North Cardinal St.
Dorchester Center, MA 02124
Work Hours
Monday to Friday: 7AM - 7PM
Weekend: 10AM - 5PM
Address
304 North Cardinal St.
Dorchester Center, MA 02124
Work Hours
Monday to Friday: 7AM - 7PM
Weekend: 10AM - 5PM

Well leakage has always been a significant challenge in the petroleum engineering field. It not only causes drilling resource losses and increased costs, but can also trigger complex conditions such as well collapse and blowouts, increasing non-productive time and even severely impacting reservoirs and the environment. Statistics show that global losses due to well leakage reach $4 billion annually, increasing drilling non-productive time by approximately 10%, negatively impacting the sustainable development of the petroleum industry.
Currently, common plugging methods include stop-drill plugging and drilling-while-plugging. Stop-drill plugging is mainly for wells prone to well collapse, stuck holes, and severe leakage. Due to the need for drill bit disassembly, the plugging process is cumbersome, time-consuming, and ultimately has limited effectiveness. Drill-while-plugging refers to the process where plugging material enters the leakage channel under pressure differential during drilling, forming a sealed plug and reducing the leakage rate. This method can address well leakage in real-time during drilling, effectively reducing losses caused by leakage and improving drilling efficiency and reducing costs.
Drill-while-plugging materials are mainly divided into physical and chemical types. Physically-based plugging materials used while drilling (walnut shells, fibers, mica sheets, etc.) have advantages such as ease of use and non-toxicity. However, due to the varying pore sizes of the leakage channels and the difficulty in matching them with the plugging materials, they are prone to “sealing off” phenomena, resulting in poor plugging effects and low success rates for one-time plugging. Chemically-based plugging materials used while drilling (polymer gel particles) have the advantages of absorbing water and swelling, deforming under pressure, and being able to penetrate deep into the leakage channels. However, currently commonly used water-absorbing and swelling gel particle plugging materials have problems such as weak pressure resistance, and the particles do not adhere to each other and are easily dispersed and carried out of the wellbore by fluctuating pressure, causing secondary leakage.
Most drilling plugging gel particles are made from acrylamide (AM) or acrylic acid (AA). AM contains amide groups in its structural units, exhibiting high chemical reactivity, readily forming hydrogen bonds, and easily grafting or cross-linking with other materials. Acrylates have good water absorption and swelling properties and water retention, enabling them to plug leakage channels. However, these gels lack self-healing properties; they only plug the leak layer through mutual compression and expansion, leaving gaps and resulting in poor plugging effectiveness or even easy backflow due to poor retention capacity. Hydrophobic associating polymers, due to the presence of hydrophobic monomers, allow intramolecular and intermolecular nonpolar groups to approach and contact each other, forming elastic recoverable micelles, thus generating dynamic association and giving them a certain degree of self-healing ability. He Meiling et al. prepared a self-healing hydrophobic associating polyacrylamide hydrogel using AM and octadecyl methacrylate (SMA). This gel can heal at 70℃, and its healing mechanism relies on the interaction between the hydrophobic monomers and solubilizers on the gel cross-section to repair the cut. Yuan et al. introduced xanthan gum as a physical crosslinking agent into hydrophobic associative polymers, which improved the mechanical properties and self-healing ability of the gel. Compared with xanthan gum, chitosan has a higher melting point, and its amino groups undergo protonation under acidic conditions, becoming positively charged, allowing it to bind tightly to the rock wall through electrostatic interactions.
To further improve the self-healing sealing performance of the drilling plugging gel, this paper uses AM and AA as hydrophilic monomers, SMA as a hydrophobic monomer, and chitosan as a physical crosslinking agent to prepare a self-healing drilling plugging gel at high temperatures, and studies its expansion, rheology, self-healing, and sealing properties.
1. Gel Preparation :
The gel was synthesized using a one-pot method. First, 2 g of CTAC was added to a 1% NaCl aqueous solution and stirred at room temperature for 30 min. Then, 0.5 mL of SMA was added, and stirring continued for 2 h until a homogeneous and transparent solution was formed. Next, 9.5 g of AM, 0.1 g of CS, and 1 mL of AA were added to the above solution, and stirring was carried out until CS was completely dissolved. Initiator KPS was added, and stirring was carried out for 10 min. Finally, the solution was placed in a mold and reacted at 70 °C for 6 h to obtain a 1.5 mol/L self-healing gel.
To study the effect of the total concentration of gel monomers on gel performance, gels with three different molar concentrations of 0.5, 1.5, and 2.5 mol/L were prepared. As the molar concentration of monomers decreased, the synthesized gel gradually softened and its strength gradually decreased. However, if the molar concentration was too high, the gel would become brittle due to its overly dense internal network structure. Therefore, 1.5 mol/L was selected as the final performance evaluation gel-while-drilling plugging agent, denoted as AACS.
2. Gel Synthesis Principle
As is well known, acrylic acid is a commonly used monomer for hydrogels. Its solution is acidic, which can deprotonate the amino groups in chitosan to form ammonium salts, and form a reversible physical cross-linking network with the long chains of acrylic acid polymers through electrostatic interaction. However, acrylic gels absorb water too quickly and expand too much before entering the leakage layer, blocking the annulus. In order to slow down the gel expansion rate, the coating method, the grafting of hydrophobic monomers method, and the non-aqueous carrying method are commonly used at home and abroad. Among them, the coating method is too complicated and easy to be unevenly coated, making it difficult to industrialize; the non-aqueous carrying method affects the final expansion ratio of the gel due to the formation water concentration problem. Therefore, the grafting of hydrophobic monomers method was finally selected to control the gel expansion rate [24]. The schematic diagram of the preparation process of self-healing gel and the non-covalent bond interaction between the gel network is shown in Figure 1.
Figure 1. Schematic diagram of the self-healing gel preparation process.
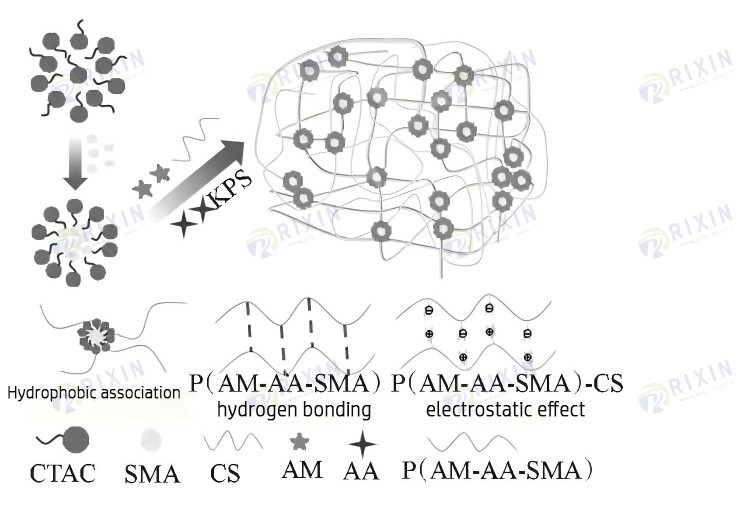
3. Evaluation of Gel Performance
3.1 Swelling
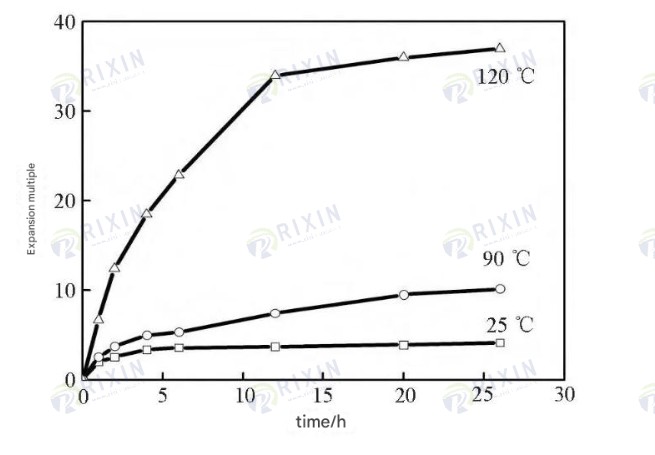
AACS The water absorption and swelling ratios of the gel at room temperature, 90℃, and 120℃ are shown in Figure 5. With increasing swelling time, the gel swelling rate first increases, then decreases, and finally tends towards equilibrium. After swelling equilibrium, the gel swelling equilibrium value increases with increasing temperature, reaching a swelling ratio of 37 times at 120℃. This is because, within the gel’s tolerable temperature range, the activity of hydration groups in the gel structure, such as amide groups, gradually increases with increasing temperature, giving the gel stronger water absorption capacity. In addition, the dynamic movement within the gel network also gradually becomes more active with increasing temperature, allowing the gel structure to retain more water.
Figure 5. Expansion factor of gel at different temperatures and times
3.2 Self-Healing Properties
Gel particles (10%) were added to deionized water and left to stand at 90 and 120°C for a period of time to observe their self-healing phenomenon. At 90°C, the gel formed by complete self-healing of the gel particles exhibited a certain degree of toughness; at 120°C, the gel particles also self-healed to form a gel whole, but the overall toughness was poor.
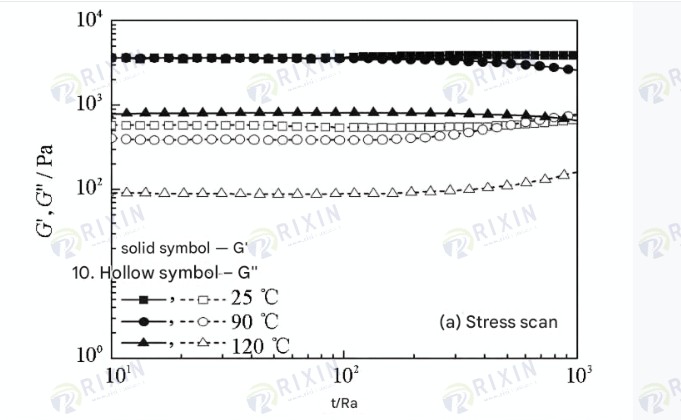
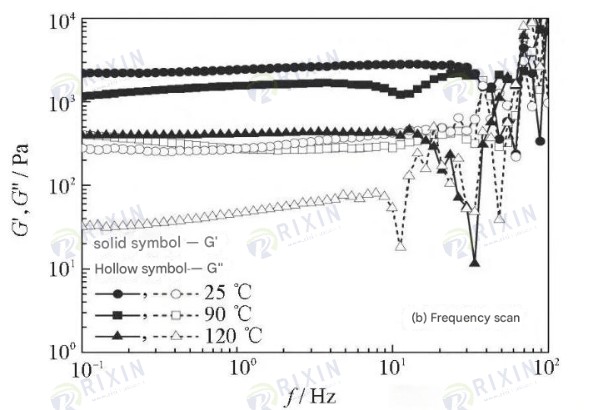
Viscoelasticity tests were performed on the healed gel using a Hacker rheometer, and the results are shown in Figure 7. It can be seen that the gels healed at high temperatures all exhibited good solid-like properties (G’>G”); the higher the ambient temperature, the significantly lower the strength of the healed gel. During stress scanning, the G’ and G” of the self-healing gel at 90°C were 3500 and 400 Pa, respectively, which are almost the same as the viscoelasticity of the gel at room temperature; the G’ and G” of the self-healing gel at 120°C were approximately 800 and 90 Pa, respectively. This shows that high temperature significantly affects the strength of the healed gel. During the frequency scanning process, the gel exhibits significant fluctuations at high frequencies, with a stable frequency between 0.1 and 10 Hz. As the healing temperature increases, the gel’s G’ decreases from 2600 Pa to 1600 Pa and then to 430 Pa.
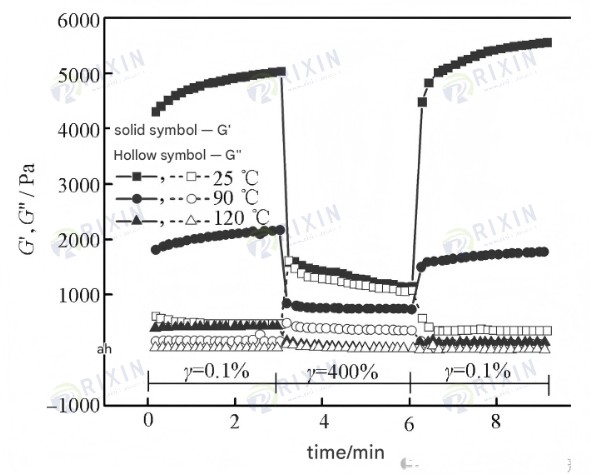
Step strain tests were performed on the three gels using a Hacker rheometer, and the results are shown in Figure 8. It can be seen that during small-amplitude oscillations (γ=0.1%), the G’ values of the three gels were approximately 4700, 2100, and 450 Pa, respectively, all higher than G”, exhibiting solid-like properties. During large-amplitude oscillations (γ=400%), the G’ values of the gels were approximately equal to G”. However, when the amplitude was reduced again to 0.1%, the G’ and G” values of the gels at 25℃ could recover to their initial values, with the structural recovery rate even exceeding 100%, and the recovery time was less than 1 min. This is mainly attributed to the non-covalent bonding in the gel structure, specifically including micelles formed by hydrophobic association between hydrophobic chains and surfactants, electrostatic interactions between polymer long chains and chitosan long chains, and hydrogen bonding between polymer long chains. These interactions enable reversible physical cross-linking between gels, allowing for cross-linking healing upon re-contact after damage. However, the structural recovery rates of the self-healing gel at 90℃ and 120℃ were only 83% and 11%, respectively, which again shows that high temperature has a significant impact on the healing strength of the gel.
Figure 8. Viscoelastic curves of gel under low strain, high strain, and low strain.
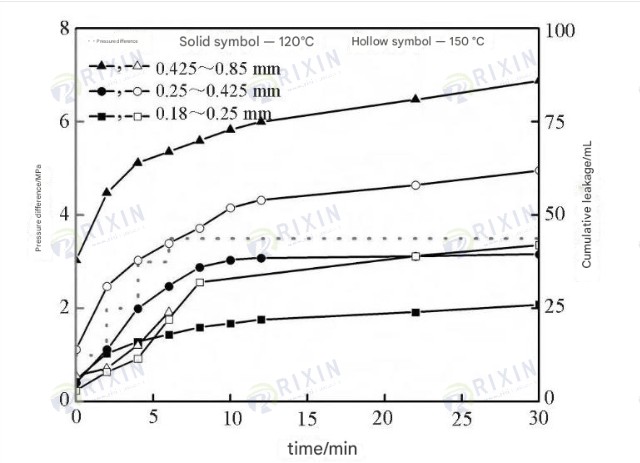
3.3 Sealing
Performance After the sealing experiment, a relatively dense sealing layer was observed to form on the surface of the sand bed. Dividing it into two parts and reassembling them, then allowing it to stand at room temperature for 1 day, self-healing was observed in the sealing layer, further demonstrating the self-healing function of the gel. The gel sealing results show that the base slurry with 2% gel particles can seal sand beds with quartz sand particle sizes ranging from 0.25 to 0.425 mm and 0.18 to 0.25 mm, with a temperature resistance of up to 150℃. The leakage amounts under 3.5 MPa for 30 minutes were 62 and 42 mL, respectively. The 0.425–0.85 mm sand bed showed puncture leakage under 3 MPa and 150℃ conditions, with a leakage amount of 86 mL under 120℃ and 3.5 MPa conditions for 30 minutes.
3.4 Gel Self-Healing and Sealing Mechanism
The sealing mechanism of self-healing gel particles in porous media and fractures is similar to that of polymer gel particles. Combined with rheological studies of the gel, it was found that the self-healing gel possesses excellent toughness and compressive deformation properties. Compared with rigid particles, this gel can penetrate leakage channels with smaller pore sizes and continuously deform and migrate under pressure differentials, thus exhibiting high adaptability to leakage channel sizes. Compared with pure gel particles, the developed gel has self-healing properties. A large number of gel particles migrate, accumulate, and compress within the leakage channels, resulting in close contact between gel particles and between gel particles and the formation. Under multiple non-covalent bond interactions (hydrogen bonding, electrostatic interactions, and hydrophobic association), self-healing between gel particles and tight connection with the formation are achieved, forming a strong and tough gel sealing layer, improving the sealing and retention capacity of the gel in the leakage channels. A schematic diagram of the self-healing gel sealing process in the leakage layer is shown in Figure 10.
Figure 1.0 Mechanism of self-healing gel plugging during drilling

4. Conclusion
Leveraging the excellent viscoelasticity of polymerized gels, the self-healing ability enhanced by chitosan, and the retention and swelling capacity enhanced by hydrophobic monomers, a dual-network structure non-covalently crosslinked self-healing gel was prepared by polymerizing chitosan as a physical crosslinking agent with acrylamide, acrylic acid, and octadecyl methacrylate. This gel exhibits excellent rheological and mechanical properties and high-temperature self-healing performance, laying a solid foundation for achieving high-pressure, high-sealing properties in drilling plugging agents. When the gel particle mass fraction reaches 2%, it can meet the drilling plugging requirements of porous formations with a mesh size of 20 or larger.