Address
304 North Cardinal St.
Dorchester Center, MA 02124
Work Hours
Monday to Friday: 7AM - 7PM
Weekend: 10AM - 5PM
Address
304 North Cardinal St.
Dorchester Center, MA 02124
Work Hours
Monday to Friday: 7AM - 7PM
Weekend: 10AM - 5PM

1. Aqueous solution polymerization
Among existing acrylamide polymerization technologies, aqueous solution polymerization is the most classic and widely used method, and it is the core technology for preparing polyacrylamide. This method promotes the polymerization reaction of monomers by directly adding an initiator in an aqueous medium or by using radiation, photoinitiation, electroinitiation, etc. After dehydration, granulation, drying, and pulverization, the product is obtained as a powder, which typically has a solid content of around 30%.
Aqueous solution polymerization is the simplest method. It typically only requires adding acrylamide monomer to the aqueous phase in an anaerobic environment, adding an initiator, and continuously stirring under controlled temperature to obtain the product. Because aqueous solution polymerization does not require an oil phase and can react only in the aqueous phase, it is simple to operate, low in cost, and requires simple equipment. The viscosity of the product is generally low, and the molecular weight of the polymer is generally below 7 million. Therefore, the thermal conduction within the system is relatively simple, heat dissipation is fast, and the reaction is less prone to explosive polymerization if the temperature is well controlled. However, the molecular weight of the product from this reaction method is generally not very high.
Polyacrylamide products synthesized by aqueous solution polymerization are generally in powder form. While easy to store and transport, they are difficult to dissolve, mainly due to the difficulty in powder dispersion and uniform, continuous injection. This is especially problematic in high-salinity brine or backwash water, where clumping and blockages can occur, leading to increased pump pressure and safety hazards. This method is unsuitable for fracturing fluid systems requiring high viscosity.
2. Suspension Polymerization:
Suspension polymerization uses an oil-soluble initiator. Because this initiator is insoluble in water, it forms monomer-containing aqueous droplets under the action of a suspending agent for polymerization. The system consists of monomer, oil-soluble initiator, water, and a suspending agent.
During polymerization, the suspending agent is added and the mixture is thoroughly stirred to uniformly disperse the monomer into small droplets. The oil-soluble initiator initiates the polymerization reaction. After several hours of temperature-controlled reaction, bead-like products are obtained. These products are repeatedly washed with organic solvents and dried to obtain bead-like products or powders with uniform particle size distribution.
This polymerization method, like aqueous solution polymerization, has rapid heat dissipation and is less prone to explosive polymerization. However, improper dispersion and dosage control of the initiator can cause self-accelerating reactions, terminating polymerization and resulting in excessively short molecular chains. Synthesizing high molecular weight polymers is relatively difficult. Furthermore, the additives in the reaction system are complex, making it easy to introduce impurities.
3. Overview of Reverse Emulsion Polymerization
(1) Introduction to Reverse Emulsion Polymerization
The reverse emulsion polymerization system mainly consists of AM, functional monomers for modification, initiators, emulsifiers, an aqueous phase, a non-polar continuous phase, and other auxiliaries. The continuous phase is the oil phase, generally saturated hydrocarbons, often white oil. The dispersed phase is the aqueous phase, in which the monomers to be polymerized are dissolved to form the dispersed phase. High-speed mechanical stirring and the addition of emulsifiers emulsify the two phases, producing a stable pre-emulsion that uniformly disperses the water-in-oil droplets, providing a site for the polymerization reaction.
Polymerization initiation can be carried out using chemical reagents or radiation methods, the latter including electric, light, and radiation initiation, but these initiations have large energy fluctuations. The former, chemical initiating reagents, come in two types: inorganic and organic. The main difference lies in the water-soluble redox initiation system and the oil-soluble azo reagent, typically AIBN. AIBN has a high initiation temperature and can explode at high temperatures, posing a safety hazard and leading to instability in the polymerization reaction system. Redox initiation systems, on the other hand, require
lower initiation temperatures, starting at around ten degrees Celsius to release free radicals. Low-temperature initiation easily yields high molecular weight products, and the reaction system is mild and does not explode. However, it is susceptible to oxygen, so an inert gas must be introduced to purge oxygen before initiation, and the entire reaction process is protected by aeration, typically using nitrogen.
The emulsion products from reverse emulsion polymerization can be made into granules for continued use without affecting performance, significantly extending product shelf life. Emulsion-type polyacrylamide is easy to dissolve and can be mixed online throughout the process. Using an online mixing vehicle, after injecting the pre-fracturing fluid to create a fracture, subsequent increases in fracturing fluid viscosity can be achieved by gradually injecting the emulsion or crosslinking agent, allowing for adjustments within the viscosity range. This significantly reduces pump pressure, saves raw materials, and reduces safety hazards.
(2) Overview of Reverse Emulsion Polymerization
Reverse emulsion polymerization is a free radical polymerization process initiated by the decomposition of free radicals by an initiator. It mainly consists of four elementary reactions: chain initiation, chain propagation, chain transfer, and chain termination. Chain transfer leads to the formation of branches, affecting the formation of macromolecules. The currently accepted polymerization method is micellar nucleation, which can be divided into four stages: dispersion stage, latex particle formation stage, latex particle growth stage, and polymerization completion stage.
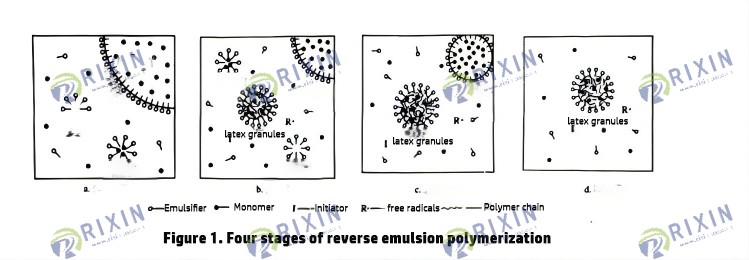
Based on the above survey and mechanism analysis of various polymerization methods, it can be seen that the products obtained by aqueous solution polymerization and suspension polymerization cannot meet the requirements of online mixing throughout the entire process. The reverse emulsion polymerization method is adopted. The polymerization process is stable and mild with no safety hazards. Its emulsion products have excellent rapid swelling characteristics and can be continuously mixed online throughout the entire process of fracturing and transformation construction. It can be used immediately after preparation, saving material costs.