Address
304 North Cardinal St.
Dorchester Center, MA 02124
Work Hours
Monday to Friday: 7AM - 7PM
Weekend: 10AM - 5PM
Address
304 North Cardinal St.
Dorchester Center, MA 02124
Work Hours
Monday to Friday: 7AM - 7PM
Weekend: 10AM - 5PM
Editor’s Note: In the previous chapters, with the aid of the “Eye of Science”—the CNGTX Spinning Drop Tensiometer—we successfully captured the ultra-low interfacial tension (Ultra-Low IFT) signals, which were once invisible yet crucial, down to the minutest detail. We have established the central role of interfacial tension as the driving force for breaking the oil-water interface and understood its dialectical role in avoiding microemulsion formation. However, seeing the battlefield clearly is only the first step toward victory. When faced with thousands of potential chemical reagent candidates, raw data does not automatically translate into solutions for complex field problems. The real challenge facing engineers lies in: How do we use this microdata, precise to 10⁻³ mN/m, to construct a winning tactical system? How do we translate every minute rotation within a laboratory capillary into the efficient separation of millions of tons of oilfield produced fluid? In this final chapter, we leave the observation deck and enter the “Command Center” of formulation design, revealing how engineers utilize this scientific ruler—from massive screening to precise compounding—to forge the “Champion Demulsifier” capable of delivering a decisive blow.
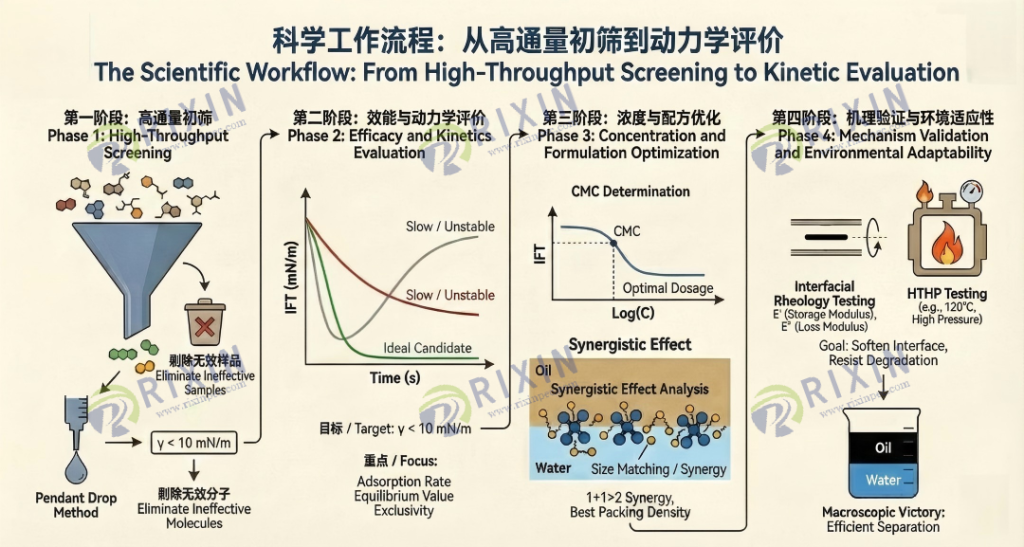
4.1 The Scientific Workflow: From High-Throughput Screening to Kinetic Evaluation
Based on interfacial tension testing, a complete R&D workflow from primary screening to fine-tuning optimization can be constructed. This is not merely a stack of testing steps, but a logically rigorous, progressive funnel screening system. Each phase has its specific physical criteria and tactical objectives, designed to rapidly lock onto the most promising molecular structures at the lowest experimental cost. This workflow is designed as four key stages, each acting as a precisely engineered checkpoint, ensuring that only molecules with true high-performance potential can pass through.
Phase 1: High-Throughput Screening—The “Filter” for Eliminating Ineffective Samples
The objective of this phase is extremely clear: to rapidly eliminate ineffective samples from a large pool of candidates. In the early stages of industrial R&D, we often face a vast library of compounds, including polyethers of various molecular weights and block ratios, as well as modified resins and sulfonates. At this point, pursuing extreme measurement precision is not the primary task; efficiency is key.
In terms of methodology, the Pendant Drop Method or automatic force tensiometers are typically used to measure the equilibrium interfacial tension at a standard concentration (e.g., 50 ppm) at ambient temperature. These methods are simple to operate and highly automated, making them suitable for handling large batches of samples. Rather than running the expensive spinning drop tensiometer at full capacity during the preliminary screening phase, it is better to first use the pendant drop method for a “mass audition”. Through automated liquid handling workstations, hundreds of samples can be tested in parallel within a short period, drastically reducing the R&D cycle.
The setting of criteria is crucial. We typically set an empirical threshold (e.g., γ< 10 mN/m). This value is not chosen arbitrarily but is based on the statistical results of extensive historical data. Any sample failing to reduce the crude oil/water interfacial tension below this threshold usually indicates insufficient surface activity or an inability to form an effective adsorption layer at the oil-water interface. Such samples are directly discarded and do not enter the next round of more time-consuming tests. The cruelty of this phase lies in its intolerance of “mediocrity”—if a sample cannot even achieve basic tension reduction, it absolutely cannot shoulder the heavy responsibility of destroying the robust interfacial film.
Phase 2: Efficacy and Kinetics Evaluation—A Battle of Speed and Passion
Methodologically, we use the Pendant Drop or the more precise Spinning Drop method to record dynamic tension curves (γ vs. t). This curve contains a wealth of information and serves as a “videotape” of the demulsifier molecules’ various “actions” at the interface. It is not merely a static value but a time-dependent function, reflecting the entire process of molecular diffusion from the bulk phase, subsurface adsorption, to interfacial rearrangement.
We calculate the decay slope from t=0 to t=teq. The larger the slope and the steeper the curve, the faster the diffusion speed of the demulsifier molecules from the bulk phase to the interface. This usually corresponds to a smaller molecular weight or a more optimized hydrophilic/hydrophobic structure, allowing it to rapidly seize the interfacial position in turbulence. This is a vital indicator for short-process operations(such as offshore oilfields). In diffusion-controlled kinetic models (such as the Ward-Tordai equation), the adsorption rate is directly correlated with the molecular diffusion coefficient and bulk concentration.
Compare the final γeq. Although we observed this value in primary screening, in this phase, we need more precise readings, even distinguishing the difference between 0.1 mN/m and 0.05 mN/m. A lower equilibrium value implies stronger interfacial displacement capability. High-efficiency demulsifiers can typically reduce tension by 1-2 orders of magnitude; this depth of tension reduction is a prerequisite for destroying the mechanical strength of the asphaltene film.
Observe for any “rebound” phenomena. If the tension curve rebounds after declining, this is often an extremely dangerous signal. It may imply competitive adsorption, molecular rearrangement after multilayer adsorption, or even that the demulsifier molecules are being “counter-attacked” by naturally active substances at the interface or undergoing complex reactions. This instability can lead to turbidity in the water phase after dehydration or re-thickening of the emulsion layer in the field.
Having identified the best molecules, the next questions are: How much to use? How to blend them? This is the task of the third phase: determining the optimal dosage concentration (CMC) and compounding ratios. This step is directly related to the project’s economics (OPEX) and the final treatment effectiveness.
n the CMC determination, we plot the “Interfacial Tension-Concentration” logarithmic curve. The inflection point of the curve is the Critical Micelle Concentration (CMC). This is a balance point between economics and technology. Typically, the optimal dosage is around the CMC. If the dosage is far below the CMC, the molecules cannot cover enough interfacial area, leading to incomplete demulsification; if the dosage is far above the CMC, it not only causes massive waste of chemicals but may also trigger “reverse stabilization”—where excess surfactant molecules form micelles in water that solubilize oil droplets, or form reverse micelles in oil that solubilize water droplets, making the emulsion even more stubborn. This is the microscopic physical essence of why field engineers often say, “adding too much demulsifier prevents dehydration”.
In synergistic effect analysis, we measure the IFT of compound systems (e.g., polyether + sulfonate, or combinations of polyethers with different EO/PO ratios) at different ratios. Our goal is to find the lowest tension point where“1+1>2”. This point usually corresponds to the optimal packing density of molecules at the interface. For example, large-molecule polyethers may cover the interface like a large umbrella, while small-molecule sulfonates fill the gaps under the umbrella; the two work closely together to thoroughly squeeze natural asphaltenes out of the interface.
Phase 4: Mechanism Validation and Environmental Adaptability—Survival Testing at the Limit
In the final phase, we need to validate the demulsification mechanism and simulate extreme field conditions. This is akin to the final combat exercise for “special forces”.
Interfacial Rheology Testing is the core weapon of this phase. We measure the Interfacial Modulus by oscillating the droplet. An efficient demulsifier must not only lower tension but also significantly reduce the interface’s Viscoelasticity, i.e., “soften” the interfacial film. If the tension is low but the modulus is high, it indicates the interface is still elastic like rubber, and droplets will merely bounce off each other upon collision rather than merging. Demulsification can only truly occur when the modulus also drops significantly, making the interfacial film as fragile as glass. Through the CNGTX800 spinning drop dilatational rheometer, we can measure the storage modulus (E’) and loss modulus (E”) of the interface, thereby quantitatively assessing the brittleness of the film.
At the same time, high-temperature and high-pressure testing must be conducted. Using equipment like the CNGTX701 high-temperature, high-pressure spinning drop tensiometer, IFT is measured at simulated reservoir temperatures (e.g., 120°C) and actual salinity. This step is to rule out the risk of failure due to thermal degradation or salting-out effects. Many demulsifiers that perform excellently at room temperature will suffer from Cloud Point precipitation or hydrolysis failure once they enter a high-temperature, high-salinity environment. Only after passing this test is the formulation truly finalized.
Having mastered the screening workflow, we need to delve into the molecular level to understand why certain structures are more effective than others. This is not only an explanation of phenomena but also a guide for future molecular design. Minute changes in molecular structure, such as increased chain length, the introduction of branches, or the substitution of functional groups, can trigger a butterfly effect on macroscopic interfacial behavior.
Structure-Activity Relationship of Polyether Demulsifiers: The Art of Balance
Currently, mainstream non-ionic demulsifiers are mostly block copolymers ofethylene oxide (EO)andpropylene oxide (PO). IFT testing reveals a profound dialectical connection between theEO/PO ratioand interfacial behavior.
Hydrophilicity is mainly provided by the EO segment, which determines the molecule’s water solubility and diffusion speed in the aqueous phase. If the EO content is too high, the demulsifier acts like a “homebody,” tending to stay in the aqueous phase rather than going to the oil-water interface front line. Lipophilicity is provided by the PO segment, endowing the molecule with interfacial adsorption capability and penetrating power into the asphaltene film. If the PO content is too high, the molecule may dissolve completely in the oil phase, also failing to accumulate at the interface.
Through precise IFT measurement, we can find that when the EO/PO ratio reaches a specific Balance Point, the interfacial tension is lowest, and the interfacial adsorption amount is maximized. This balance point is the famous Hydrophile-Lipophile Balance (HLB). Furthermore, molecular topology is crucial. Research indicates that branched structures (such as multi-armed star polymers or dendrimers) typically possess stronger interfacial displacement capabilities than linear structures. This is manifested as a faster rate of tension reduction on the dynamic tension curve—star-shaped molecules act like a multi-clawed anchor; once they grab the interface, they are hard to dislodge and can more effectively disrupt the dense packing of asphaltenes. For instance, Dendritic Polyethers, due to their high geometric symmetry and numerous terminal functional groups, have demonstrated superior interfacial activity and demulsification efficiency compared to linear polyethers.
Disruption Mechanisms of Hydrogen Bonds and π-π Stacking: Scalpels at the Micro Level
Recent research has introduced molecular dynamics simulations combined with IFT testing, revealing how demulsifiers act like scalpels to precisely cut the connections between asphaltenes. Asphaltene molecules tend to aggregate via “island” or “archipelago” models, forming robust interfacial films. High-efficiency demulsifiers (such as nitrogen- and oxygen-containing compounds) can insert themselves into asphaltene aggregates by forming stronger hydrogen bonds or π-π interactions.
This mechanism can be vividly described as a“Trojan Horse”. Demulsifier molecules first disguise themselves with structures similar to asphaltenes, infiltrating the interfacial film, and then “break” the original π-π stacking network of asphaltenes through stronger non-covalent interactions. Once the network is broken, the sturdy film disintegrates.
Synergistic effects play a huge role in this mechanism. For example, blending fluorinated polyethers (possessing extremely high surface activity) with ordinary polyethers. IFT data shows that the fluorinated component can reduce tension extremely rapidly, acting as the “vanguard” to tear open the defense line; while the ordinary polyether follows up, thoroughly destroying the film structure through hydrogen bond reconstruction, acting as the “main force”. Research has found that fluorinated polyethers, due to the extremely low surface energy and high stability of the C-F bond, can rapidly reduce oil-water interfacial tension and possess shear resistance, preventing secondary emulsification. This“dual-sword”effect can only be quantitatively captured through precise dynamic IFT and rheology testing, explaining why certain compound formulations far outperform single components.
Introduction of Magnetic and Green Nanomaterials
Beyond traditional organic molecules, novel functionalized nanomaterials are also emerging. Magnetic demulsifiers(such as Fe₃O₄ nanoparticles) can not only achieve rapid separation via an external magnetic field but can also be recycled and reused. Studies show that Fe₃O₄ nanoparticles grafted with fluorinated polyethers not only inherit the low surface energy properties of fluorides but also possess magnetic responsiveness, maintaining a dehydration efficiency of over 95% across multiple cycles. The introduction of such materials fundamentally challenges the traditional concept of demulsifiers as “single-use” chemicals.
4.3 Market Trends and Future Outlook
With increasingly stringent global environmental regulations and the advancement of the digital wave, the demulsifier market is undergoing profound transformation. This market is no longer just about the trading of chemicals but is evolving towards high-tech, green, and intelligent directions.
According to authoritative forecasts, the global demulsifier market size is projected to grow significantly from approximately $2.5 billion in 2025 to over $3.7 billion by 2035, with a Compound Annual Growth Rate (CAGR) of about3.5% to 3.8%.
This growth is primarily driven by the following factors:
Deterioration and Heaviness of Crude Oil: With the depletion of easily accessible oil fields, global crude oil extraction is increasingly shifting towards heavy oil, bitumen, and complex emulsions produced by Enhanced Oil Recovery (EOR). These crude oils contain more asphaltenes and resins, resulting in more stable emulsions, which drastically increase the demand and performance requirements for high-efficiency demulsifiers. The Middle East and Africa (MEA) region, possessing vast heavy oil reserves, is expected to capture nearly 40% of the market share.
Pressure from Environmental Regulations:The strict enforcement of produced water treatment standards by governments (such as China’s“Double 20” standard, i.e., oil content and suspended solids both less than 20 mg/L) compels oilfield companies to use higher-performance demulsifiers to ensure discharge compliance.
Rise of Emerging Markets: The Asia-Pacific region(especially China and India), with its industrialization and increasing energy demand, is becoming the fastest-growing regional market.
The Rise of Green Demulsifiers
Although traditional polyether demulsifiers are efficient, they are often difficult to degrade and may contain aromatic solvents, posing a threat to the marine environment. Therefore, green demulsifiers—bio-based and biodegradable demulsifiers—are becoming research hotspots.
Current research primarily focuses on plant oil-modified products. For example, modifying feedstocks like coconut oil, soybean oil, cashew nut shell liquid, or corn oil to synthesize novel demulsifiers. These bio-based demulsifiers have shown impressive performance in laboratories; some formulations (such as modified coconut oil demulsifier COSD) can achieve 100% oil-water separation in just 30 minutes at 70°C, while exhibiting excellent biodegradability.
For these novel molecules, IFT testing faces new challenges. Bio-based molecules often have complex compositions and significant batch-to-batch variations. This requires our measurement instruments to possess higher robustness and adaptability, capable of maintaining measurement accuracy in complex biological matrices, and establishing new evaluation standards to balance the contradiction between“environmental friendliness” and “activity”.
Customization and Intelligence: AI-Driven Formulation Design
The biggest transformation in the future will come from Artificial Intelligence (AI). Traditional “trial-and-error” methods are inefficient and rely on experience. Utilizing AI and Machine Learning (ML), combined with big data generated by high-throughput IFT testing, it will become possible to predict the optimal demulsifier formulation for unknown crude oils.
We can train Multilayer Perceptron (MLP) or Artificial Neural Network (ANN)models by inputting the physical properties of crude oil (e.g., density, viscosity, asphaltene content, water cut) and the molecular parameters of candidate demulsifiers (e.g., EO/PO ratio, molecular weight, HLB value) to output predicted IFT values and Water Removal Efficiency (WRE). Research indicates that MLP models exhibit extremely high accuracy in predicting dehydration efficiency, with average absolute relative errors as low as0.84%. This is like equipping chemists with a super brain capable of screening millions of virtual formulations in seconds, shortening the R&D cycle from weeks to hours.
Furthermore, sensor technology for real-time online monitoring of interfacial tension is expected to be integrated into oilfield automation systems, enabling closed-loop control of dosage—when crude oil properties fluctuate, the system automatically adjusts the dosage, always maintaining it at the optimal CMC point, ensuring both dehydration effectiveness and cost savings. This“Smart Oilfield Chemicals Management”mode (such as the SMART View system) is becoming the new industry standard.
4.4 Conclusion
In summary, oil-water interfacial tension testing is not merely a simple physical parameter measurement; it is the core bridge connecting the molecular structure design of demulsifiers with macroscopic dehydration performance.
It plays three critical roles:
As a Screening Tool:It provides a rapid, quantitative elimination mechanism, with dynamic IFT being key to evaluating adsorption kinetics.
As a Mechanistic Probe:Combined with interfacial rheology, it reveals the dialectical relationship between “lowering tension” and “breaking the film,” helping researchers avoid the microemulsion trap.
As an Optimization Method: Through CMC determination and synergistic effect analysis, it guides the refined design of formulations and the economic control of field dosage.
With the proliferation of high-end measurement techniques like the Spinning Drop Method and the integration of molecular simulation tools, rational design based on interface science will thoroughly replace traditional “empirical trial-and-error,” leading demulsifier R&D into a new era of high efficiency, precision, and sustainability. In this battle of the microcosm, whoever masters the secrets of interfacial tension controls the future of oil-water separation.
From laboratory to oilfield, from minute interfacial tensions to the separation of millions of tons of oilfield produced fluid, the CNGTX Spinning Drop Tensiometer is not just a measurement instrument but a key tool driving the green and intelligent transformation of the petroleum industry. In the future, with the deep integration of AI technology and green chemistry, demulsifier R&D will enter a brand new era—precise, efficient, and sustainable.