Address
304 North Cardinal St.
Dorchester Center, MA 02124
Work Hours
Monday to Friday: 7AM - 7PM
Weekend: 10AM - 5PM
Address
304 North Cardinal St.
Dorchester Center, MA 02124
Work Hours
Monday to Friday: 7AM - 7PM
Weekend: 10AM - 5PM
Petroleum, as the lifeblood of industry, is hailed as “black gold” and plays a vital role in national and social development. With the continuous progress and development of China’s economy and society, the demand for crude oil is constantly rising. Constrained by the quality of China’s crude oil resources themselves, major oilfields have generally entered a stage of high water cut and high recovery, urgently requiring the search for new replacement resources. Low-permeability reservoirs account for as much as two-thirds of China’s total proven reserves, possessing enormous development potential and representing an important replacement resource for conventional oilfields.
Currently, conventional enhanced oil recovery (EOR) methods for low-permeability reservoirs include surfactant flooding and gas flooding. While surfactant flooding can reduce oil-water interfacial tension, wet rock surfaces, and generate foam and emulsification, it also suffers from limited oil displacement volume and low EOR efficiency. Gas flooding also faces challenges such as insufficient gas stability leading to low recovery rates and potential groundwater and surface environmental pollution. Other conventional oil displacement agents often encounter difficulties in injection and recovery when developing low-permeability reservoirs. Given these contradictions between conventional oil displacement methods and low-permeability reservoirs, new oil displacement technologies are urgently needed to support increased and stable crude oil production in my country. With the continuous development of nanomaterials technology, nanofluids, with their advantages of good injection performance, ability to penetrate deep into pore throats to activate remaining oil, and low concentration, are increasingly being applied to the exploitation of low-permeability reservoirs.
Compared to spherical nanoparticles, two-dimensional nanomaterials can be used at lower concentrations. Among them, molybdenum disulfide (MoS2) nanosheets have become a novel nano-displacement agent in recent years due to their layered structure, simple preparation method, and low production cost. Their relatively lower interfacial free energy makes them more easily adsorbed onto the oil-water interface, which has sparked increasing research interest among developers of low-permeability reservoirs.
However, in actual high-salt, low-permeability reservoir environments, the dispersion stability of nanofluids often faces significant challenges, yet research on the dispersion stability of nanofluids for oil displacement is limited. As an inorganic nanomaterial, MoS2 nanosheets exhibit poor dispersion stability in mineralized water, easily leading to aggregation and sedimentation, which is detrimental to the storage and transport of nanofluids. This also affects the dispersion and migration of MoS2 nanofluids within the formation, thereby reducing their recovery capacity. Therefore, small-scale, high-dispersion-stability, and salt-tolerant two-dimensional nanofluid displacement systems are crucial for the development of low-permeability reservoirs.
This paper first synthesizes small-scale MoS2 nanosheets using a hydrothermal method. The microstructure of these nanosheets was characterized using scanning electron microscopy and transmission electron microscopy, and their chemical composition was determined by infrared spectroscopy and X-ray diffraction. Then, the nanosheets were compounded with different surfactants. By comparing the dispersion stability, interfacial properties, and emulsifying properties before and after compounding with various surfactants, a novel nano-enhanced oil recovery system (CTAB-MoS2) was finally constructed. Finally, the enhanced oil recovery effect of CTAB-MoS2 was evaluated through core flooding experiments.
1. Sample preparation
(1) Synthesis of MoS2 nanosheets
Add 3.5 mmol MoO3, 7 mmol C2H5NS, and an appropriate amount of CH4N2O to a beaker, then add 50 mL of deionized water. Stir thoroughly at 200 r/min for 120 min, then transfer to a polytetrafluoroethylene liner in a reaction vessel. Place the liner into a high-temperature, high-pressure hydrothermal reactor, assemble the equipment, and react at 200℃ for 12 h. After the experiment, wash the product repeatedly by alternating centrifugation with deionized water and anhydrous ethanol, and freeze-dry to obtain MoS2 nanosheet powder for later use.
(2) Preparation of MoS2 nanosheet dispersion
A certain amount of MoS2 nanosheets was placed in a beaker, and deionized water or NaCl solution was added. The mixture was ultrasonically dispersed for 10 min to obtain a MoS2 nanosheet dispersion. During ultrasonication, the beaker was placed in an ice-water bath to prevent the dispersion from overheating. 0.2 g of surfactant (CTAB, SDS, AES, OPS, AEO) and 0.02 g of MoS2 nanosheets were added to a reagent bottle, followed by 20 mL of deionized water or NaCl solutions with different mineralization levels (0–3000 mg/L). After thorough ultrasonication, a MoS2 nanosheet-surfactant composite dispersion with a MoS2 nanosheet mass fraction of 0.1% was obtained. Diluting with deionized water or NaCl solution yielded MoS2 nanosheet dispersions of different concentrations.
2. Interfacial properties of the MoS2 nanosheet oil displacement system
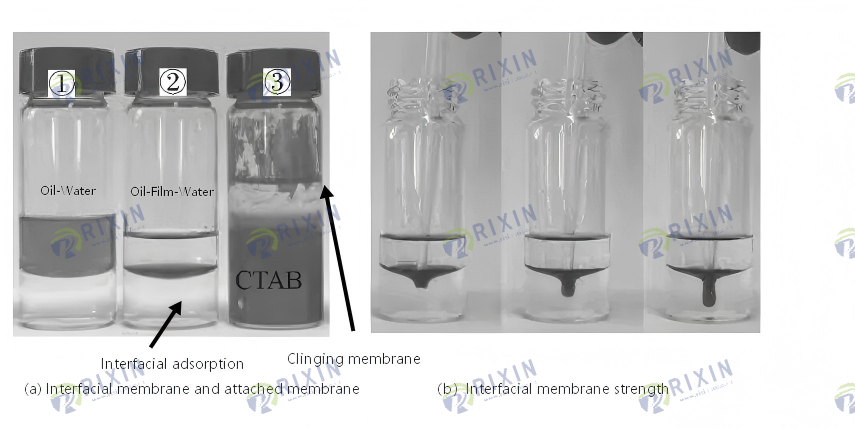
Compared with the “point-to-surface” contact between spherical nanoparticles and the oil-water interface, the sheet-like MoS2 nanosheets can spread at the oil-water interface to form a larger contact area [20]. As shown in Figure 5(a), after adding an appropriate amount of CTAB-MoS2 nanosheet oil displacement system to the cyclohexane-deionized water interface, the MoS2 nanosheets can spontaneously aggregate at the oil-water interface, resulting in obvious interfacial adsorption. After slight shaking, the CTAB-MoS2 nanosheets emulsify the oil phase to form a Pickering emulsion, and then the emulsion droplets aggregate. The distribution of CTAB-MoS2 nanosheets is concentrated at the oil-water interface and forms an interfacial film. As shown in Figure 5(b), the interfacial film can maintain a large degree of deformation without breaking under the pressure of the dropper, indicating that the formed interfacial film has good strength. At the same time, the oil-water interface density and the interfacial thickness both increase with the increase of the number of nanosheets. As can be seen from reagent bottle (3) in Figure 5(a), after the reagent bottle was shaken and left to stand in the adhesion film experiment, the black Mo S2 nanosheets spontaneously climbed up the bottle wall and formed an obvious adhesion film. This is because the aggregation of emulsion droplets increased the concentration of CTAB-Mo S2 nanosheets at the oil-water interface, which led to an increase in local surface pressure at the oil-water interface and produced the Marangoni effect [23], which pushed the CTAB-Mo S2 nanosheets from the oil-water interface to the bottle wall. The experiment measured that the interfacial tensions of the oil-water system of n-heptane-deionized water, n-heptane-nanosheets, n-heptane-nanosheets-CTAB, and n-heptane-CTAB were 27.020, 25.540, 0.175, and 0.180 m N/m, respectively.
The interfacial tension between n-heptane and the MoS2 nanosheet dispersion is similar to that between n-heptane and water, indicating that MoS2 nanosheets have virtually no effect on reducing the oil-water interfacial tension. After adding a surfactant, the MoS2 nanosheet oil displacement system can reduce the oil-water interfacial tension to 0.175 mN/m, similar to the effect of CTAB in reducing oil-water interfacial tension. This suggests that the system primarily achieves its effect through the added surfactant CTAB. In summary, the CTAB-MoS2 nanosheet oil displacement system constructed in this study exhibits good oil-water interfacial tension reduction, generates a high-strength interfacial film and demonstrates significant adhesion film phenomenon, exhibiting good interfacial adsorption activity, which is beneficial for improving the oil displacement effect.
Figure 5. Interfacial properties of MoS2 nanosheets
3. Emulsifying properties of the MoS2 nanosheet oil displacement system
The emulsifying effect of MoS2 nanosheet dispersions with different surfactants was evaluated using optical microscopy. As shown in Figure 6, each dispersion system exhibited a certain emulsifying effect. When SDS and OPS were combined, the droplet size was generally less than 100 μm, but the distribution was scattered and the number was small, resulting in poor emulsification. When CTAB and AES were combined, the number of droplets increased significantly, and the arrangement was more dense. The droplet size of the AES-combined system varied considerably, leading to unsatisfactory emulsification. The droplet size of the CTAB-combined system was generally less than 100 μm, with a more concentrated size distribution. Therefore, the CTAB-combined MoS2 nanosheet dispersion showed the best emulsifying effect. This indicates that MoS2 nanosheets and CTAB have a good synergistic effect, effectively improving both dispersion stability and emulsification. Combined with the above performance test results, the CTAB-MoS2 nanosheet oil displacement system exhibits good salt resistance, dispersion stability, interfacial activity, and emulsifying ability.
Figure 6. Emulsification effect of MoS2 nanosheet dispersions with different surfactants

4. Core oil displacement effect
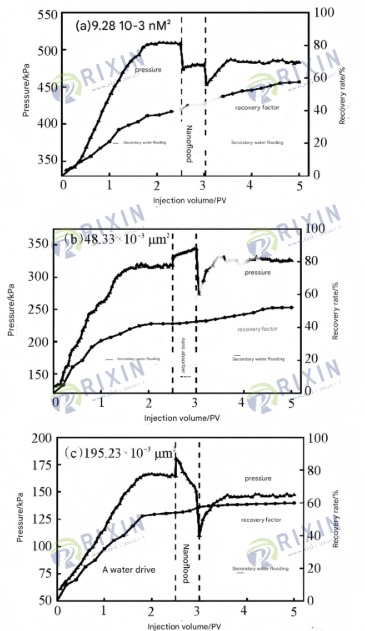
Figure 7 shows the oil displacement effect of the MoS2 nanosheet flooding system in cores with different permeabilities. With increasing core permeability, the final steady-state pressure at the end of the first waterflooding operation in each group of experiments was approximately 510, 320, and 160 kPa, respectively, showing a decreasing trend. This is because higher permeability results in greater core compaction, leading to a higher injection pressure during waterflooding. When the MoS2 nanosheet dispersion was first injected, the injection pressure at the injection end of each group of experiments increased, indicating a certain blocking effect during nanosheet injection. After injecting 0.5 PV of nanosheet dispersion, the nanoflooding end pressure corresponding to the 9.28×10⁻³ μm² and 48.33×10⁻³ μm² cores was higher than the waterflooding end pressure, possibly indicating some blocking. However, the nanoflooding end pressure in the 195.23×10⁻³ μm² core was lower than the waterflooding end pressure, indicating a larger pore throat size and no blocking effect. In core samples with permeabilities of 9.28×10⁻³, 48.33×10⁻³, and 195.23×10⁻³ μm², the MoS₂ nanosheet flooding system increased oil recovery by 9.28, 9.85, and 5.95 percentage points respectively on the basis of a single waterflood.
The MoS2 nanosheet flooding system demonstrates good enhanced oil recovery (EOR) performance in low-permeability cores. The addition of CTAB improves the dispersion stability and salt tolerance of the MoS2 nanosheets, preventing blockage in the injection section and increasing the system’s washout efficiency, thus enhancing EOR. However, in high-permeability cores, the small nanosheet size fails to seal large pores, leading to severe water channeling and ineffective activation of residual oil in low-permeability pores, resulting in poor oil displacement. Therefore, the MoS2 nanosheet flooding system is suitable for developing medium-to-low permeability reservoirs, and in high-permeability reservoirs, it needs to be used in combination with other modifiers.
Figure 7 Results of core flooding experiments with different permeabilities
5. Conclusion
MoS2 nanosheets synthesized via a hydrothermal method are small in size, with a particle size of approximately 60 nm. While MoS2 nanosheets can be stably dispersed in pure water, their dispersion stability deteriorates in mineralized water. The combination of nanosheets and the surfactant CTAB significantly improves the dispersion stability of MoS2 nanosheets in a 3000 mg/L NaCl solution; the self-stabilization time of 0.1% CTAB-MoS2 increases from less than 3 h to 12 h. The mechanism by which CTAB improves the dispersion stability of MoS2 nanosheets is as follows: CTAB adsorbs and associates on the surface of MoS2 nanosheets, causing charge reversal and increasing the absolute value of the Zeta potential, thus increasing the electrostatic repulsion between MoS2 nanosheets; the longer carbon chains of CTAB make it difficult for the nanosheets to aggregate, thereby improving the stability of the MoS2 nanosheet dispersion.
CTAB-Mo S2 exhibits excellent interfacial properties, spontaneously aggregating at the oil-water interface to form a high-strength interfacial film and a significant adhesion film, thereby significantly reducing the interfacial tension between oil and water. CTAB-Mo S2 demonstrates a certain oil displacement effect in low-permeability environments. Injecting only 0.5 PV 0.005% CTAB-Mo S2 into a low-permeability core with a permeability of approximately 50 × 10⁻³ μm² can increase the oil recovery rate by 9.85 percentage points, effectively addressing the problem of poor oil displacement effect caused by deteriorated dispersion stability during the use of nano-displacement agents.