Address
304 North Cardinal St.
Dorchester Center, MA 02124
Work Hours
Monday to Friday: 7AM - 7PM
Weekend: 10AM - 5PM
Address
304 North Cardinal St.
Dorchester Center, MA 02124
Work Hours
Monday to Friday: 7AM - 7PM
Weekend: 10AM - 5PM
Editor’s Note: Welcome to this in-depth series covering the frontiers of interfacial chemistry and petroleum engineering. Over the course of four parts, we will guide you through the microscopic world from the perspective of a multidisciplinary expert, exploring the mysteries of oil-water emulsion stability, decoding the fascinating mechanisms of demulsifiers, and discussing why only specific instrumentation can capture the critical metric of “ultra-low interfacial tension.” This series spans from thermodynamic foundations to industrial applications, exceeding ten thousand words, aiming to provide an exhaustive scientific guide for readers seeking depth. Whether you are a professional in oilfield chemistry or fluid mechanics, or simply a reader curious about the micro-physicochemical world, this series promises to provide unprecedented depth and visual insight. Let us begin with Part 1 and step into the “invisible war” occurring on the surface of micron-sized droplets.
Part 1: The Invisible Battleground — The Robust Armor of Emulsions and the Demulsifier’s Raid
1.1 Order in Chaos: Thermodynamics and the Formation of Emulsions
In the grand narrative of oil exploration and industrial processing, a minute yet stubborn physical phenomenon consistently occupies the center stage—the formation of Emulsions. When we speak of crude oil, we often overlook the fact that during extraction, it does not exist as a pure oil phase but forms a complex mixed system with formation water, silt, and various chemical additives. This is not merely a problem of fluid mechanics but a profound thermodynamic game.
From the perspective of classical thermodynamics, oil and water are immiscible. In a two-phase system, the system always tends to minimize its Gibbs Free Energy (G ). The total interfacial free energy is the product of the interfacial tension (γ ) and the interfacial area (A ). Due to the existence of oil-water interfacial tension, the system tends to separate into oil and water to minimize the contact area (i.e., dA< 0), thereby reducing the total energy of the system. However, in reality, what we often observe are extremely stable water-in-oil (W/O) or oil-in-water (O/W) emulsions. This state is thermodynamically unstable but Kinetically Stable.
This counterintuitive stability stems from two key factors: the reduction of interfacial tension and the formation of a robust interfacial film. During crude oil extraction, high-intensity shear forces(such as turbulence at pumps and choke valves) shatter the water phase into micron-sized droplets dispersed within the oil phase, drastically increasing the interfacial area. In a pure oil-water system, these droplets would rapidly coalesce again. However, crude oil is a complex “chemical soup” rich in naturally occurring surface-active substances.
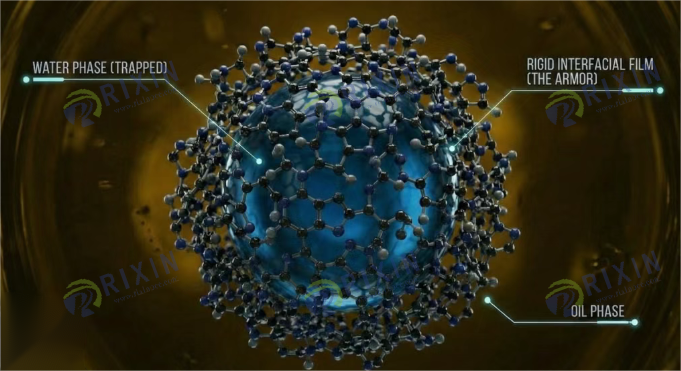
1.2 Asphaltenes and Resins: The Natural Microscopic Armor
The root of this stability lies in the naturally occurring surface-active substances within crude oil, particularly Asphaltenes and Resins, as well as minute solid particles (such as clays and silica). These substances act as natural emulsifiers.
Asphaltenes: These are the heaviest and most polar components of crude oil. They are complex macromolecules composed of polycyclic aromatic cores, aliphatic side chains, and functional groups containing heteroatoms like nitrogen, oxygen, and sulfur. Asphaltene molecules tend to form nano-aggregates through π-π stacking interactions. When these aggregates adsorb at the oil-water interface, they cross-link to form a mechanically strong, solid-like network structure.
Resins: Resins have molecular weights and polarities intermediate between the oil fraction and asphaltenes. They typically act as peptizing agents for asphaltenes, wrapping around them to prevent excessive precipitation. However, at the interface, resins act synergistically with asphaltenes, further enhancing the thickness and viscoelasticity of the interfacial film.
This film not only reduces the tension at the oil-water interface but, more critically, forms a suit of sturdy “microscopic armor.” This armor possesses significant Interfacial Viscosity and Elastic Modulus. When two droplets approach each other, this film provides powerful Steric Hindrance. Charges on the droplet surface (often derived from the dissociation of acidic/basic groups in asphaltenes) also generate electrostatic repulsion, which is particularly pronounced in low-salinity water. Therefore, the core of emulsion stability lies in the existence of this high-strength interfacial film, which imprisons water droplets like a jail, preventing them from returning to the free water phase.
1.3 Strategic Deployment of Demulsifiers: Penetration, Softening, and Unlocking
Facing this army of armor-clad “microscopic soldiers,” physical methods (such as thermal sedimentation, centrifugation separation, etc.) are often inefficient. We require a special chemical task force—Demulsifiers. Demulsifiers are essentially a class of precisely engineered surfactants, typically possessing higher Interfacial Activity than natural emulsifiers. Their mechanism of action can be vividly metaphorized as a precise “disarming” operation, a process involving complex competitive adsorption and interfacial dynamics.
The demulsification process can typically be decomposed into the following four critical steps:
Penetration and Diffusion: Leveraging their amphiphilic structure(hydrophilic head and lipophilic tail), demulsifier molecules can rapidly traverse the continuous phase (usually the oil phase) and diffuse to the interface of the dispersed phase droplets. The speed of this step determines the initial response time of demulsification.
Adsorption and Displacement: This is the crux of demulsification. Possessing higher surface activity, demulsifier molecules insert themselves like sharp knives into the dense film formed by natural emulsifiers. This is not merely physical displacement; it is a thermodynamic power play (governed by the Gibbs adsorption isotherm). Through the Marangoni Effect, demulsifier molecules create a tension gradient at the interface, driving rapid spreading and “displacing” or “solubilizing” the originally adsorbed asphaltene and resin molecules into the bulk phase.
IFT Reduction and Film Softening: The adsorption of demulsifier molecules significantly lowers the Oil-Water Interfacial Tension (IFT) and causes the originally taut, rigid droplet surface to become relaxed and pliable. If we liken a droplet to an inflated balloon, high interfacial tension means the balloon skin is very tight and resistant to deformation; lowering interfacial tension is like deflating the balloon, making it flaccid. This “softening” allows droplets to deform more easily upon collision, increasing contact area. However, lowering interfacial tension is merely the “vanguard” action of demulsification, not the whole story.
Film Rupture: Efficient demulsifiers do not just lower tension; they destroy the mechanical strength of the interfacial film. The interfacial film formed by demulsifier molecules is typically loosely structured, possesses low strength, exhibits good fluidity, and lacks viscoelasticity. This is equivalent to unlocking the “buckles” of the armor. Once the natural rigid film is replaced by the loose film formed by the demulsifier, the protective layer between droplets becomes exceedingly fragile.
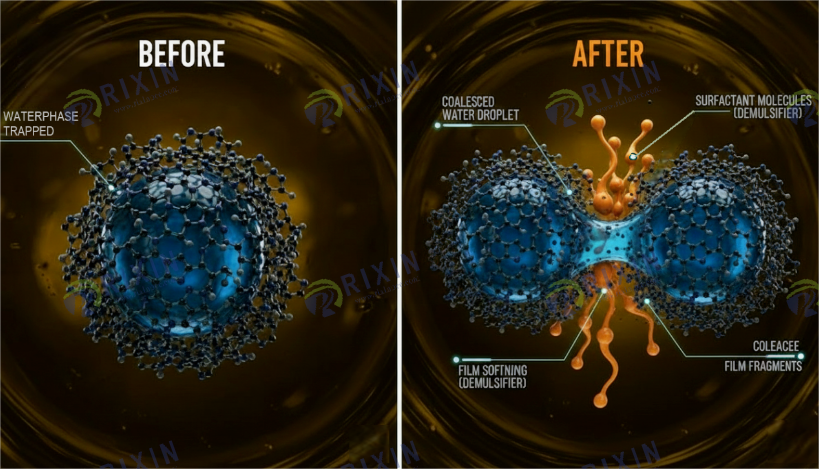
1.4 Coalescence: Victory from Micro to Macro
After the interfacial film is destroyed, Van der Waals forces begin to dominate. Droplets approach each other, and the Thin Liquid Film between them gradually thins under Drainage Pressure. Due to the loss of interfacial film strength, the film eventually ruptures, and two small droplets instantly merge into one large droplet. This process is called Coalescence.
As coalescence continues, the droplet volume increases. According to Stokes’ Law in fluid mechanics:
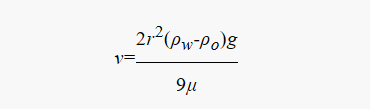
Where v is the sedimentation velocity, r is the droplet radius, Δρ is the density difference, and μ is the viscosity of the continuous phase. We can see that sedimentation velocity is proportional to the square of the droplet radius. This means that if a demulsifier can increase the droplet radius by a factor of 10, the separation speed will increase by a factor of 100! This is why microscopic interfacial modification can lead to a massive leap in macroscopic oil-water separation efficiency.
The battle at the interface is elegant yet ruthless. We have seen how demulsifiers act as strategic saboteurs, softening defenses and unlocking barriers to allow oil and water to part ways. However, a paradox remains in the scientific community: if lowering interfacial tension is the vanguard of this operation, how low must we go? Is zero tension the ultimate goal? Could tension that is too low create new problems? As we transition to Part 2, we will uncover why simply lowering the tension is not a silver bullet and explore the nuanced dialectic between tension reduction and film elasticity that dictates the success of every demulsifier formulation. We will dive into the world of data to see how invisible forces are quantified and manipulated.