Address
304 North Cardinal St.
Dorchester Center, MA 02124
Work Hours
Monday to Friday: 7AM - 7PM
Weekend: 10AM - 5PM
Address
304 North Cardinal St.
Dorchester Center, MA 02124
Work Hours
Monday to Friday: 7AM - 7PM
Weekend: 10AM - 5PM
The Bohai Sea has become one of the important bases for offshore oil and gas development in my country. The Bohai Oilfield accounts for about 70% of the total offshore oilfield production and is the largest offshore oilfield in my country [1-2]. Chemical flooding in the Bohai Sea has basically solved the polymer flooding problem for deltaic sedimentary and Class I heavy oil reserves. However, for Class II heavy oil with high underground crude oil viscosity, and under high temperature and high salinity reservoir conditions, the reservoir adaptability of chemical flooding agents still needs technical breakthroughs. Partially hydrolyzed polyacrylamide is a commonly used polymer flooding agent, mainly used for polymer flooding in onshore oilfields.
From a molecular structure perspective, conventional hydrolyzed polyacrylamide is unsuitable for use in high-temperature and high-salinity offshore oilfields. While the intramolecular and intermolecular electrostatic repulsion generated by carboxyl groups may thicken fresh water, in high-temperature brine or seawater, inorganic cations can mask this electrostatic repulsion, leading to polymer chain breakage and a reduction in hydrodynamic radius, both of which result in a macroscopic decrease in polymer viscosity. Studies have shown that with increasing system concentration, amphiphilic polymer molecules in aqueous solutions can gradually self-assemble into hydrophobic microdomains, supramolecular aggregates, and even three-dimensional dynamic cross-linked networks through hydrophobic association between hydrophobic segments. Therefore, they exhibit stronger thickening capabilities and superior temperature, salt, and shear resistance compared to conventional polymers.
Chen Minggui et al. used acrylamide (AM), sodium styrene sulfonate (PSS), methacrylic acid (MAA), and sodium 2-acrylamido-dodecyl sulfonate (C12AMPS) as reactants and prepared an amphiphilic polymer containing rigid groups by aqueous free radical polymerization. Zhang Rui et al. [10] first prepared a polymerizable surface-active functional monomer N-benzyl-N-alkylacrylamide (DTAM) using AM, bromoalkanes and benzyl bromide as raw materials, and then copolymerized DTAM, AM and sodium 2-acrylamido-2-methylpropanesulfonate (AMPS-Na) to prepare an amphiphilic polymer suitable for emulsifying and reducing viscosity of super-heavy oils. Fei Dongtao et al. used bromoalkanes, polyethyleneimine, AM, and 2-acrylamido-2-methylpropanesulfonic acid (AMPS) as raw materials and prepared an amphiphilic polymer by free radical polymerization with good temperature and salt resistance. Yang Hongbin et al. synthesized novel betaine-type amphiphilic polymers using ammonium acetate (AM) as a hydrophilic monomer, methacryloyloxyethyl-N,N-dimethylpropanesulfonate (DMAPS) as a betaine-type functional monomer, and hexadecyl dimethyl allyl ammonium chloride (C16DMAAC) as a hydrophobic monomer. The above studies on amphiphilic polymers all involved copolymerizing AM with monomers containing long-chain hydrophobic groups, while studies on copolymerizing AM with functional monomers containing both hydrophilic and hydrophobic long-chain segments are relatively few.
Other methods can also enhance the heat and salt resistance of polyacrylamide, such as increasing its molecular weight and introducing heat- and salt-resistant comonomers. High molecular weight polyacrylamide has a relatively large hydrodynamic radius, resulting in relatively high initial viscosity and residual viscosity, thereby improving the heat and salt resistance of the polymer. Introducing heat- and salt-resistant comonomers can improve the thermal and salt stability of the polyacrylamide main chain, because the introduced monomers (such as N-ethyleneamine and AMPS[16]) do not react with ions and can inhibit the hydrolysis of amide groups. In this paper, a self-made amphiphilic functional monomer (POM), AM, acrylic acid (AA), and AMPS containing polyether long-chain segments as hydrophilic groups and long-chain alkane as hydrophobic groups are copolymerized to prepare an amphiphilic oil displacement polymer by free radical polymerization. The introduction of AMPS improves the thermal and salt stability of the polymer. The introduction of hydrophilic groups in the self-made functional monomer POM improves the water solubility of the amphiphilic polymer. The association of hydrophobic groups increases the viscosity of the polymer, thereby achieving the purpose of rapid dissolution, heat and salt resistance and shear resistance of the oil displacement polymer.
1. Synthesis and Characterization of Amphiphilic Oil-Depleting Polymers
A certain mass of AM, AA, AMPS, and POM were weighed and placed in a beaker containing distilled water. The mixture was stirred and dissolved. Surfactant OP-10, urea, chain transfer agent NaH2PO2·H2O, and defoamer were added sequentially. The pH of the solution was then adjusted to 7.8–8.0 with 30% NaOH solution. The mixture in the beaker was cooled to 0°C and transferred to a heat-insulated reactor. High-purity N2 was introduced for approximately 15 min, followed by the sequential addition of initiators Na2S2O8, NaHSO3, and VAO44. Once the solution thickened, the N2 supply was stopped, and the reactor was sealed and allowed to stand. The reaction was observed using a thermometer. The system temperature reached its peak after 3–4 h. Further maturation for 8–12 h yielded an elastic polymer block. The block was pulverized using a granulator and then dried at 50–60°C for 2 h using a blower. Finally, it was pulverized again, sieved, and washed multiple times with ethanol to obtain an amphiphilic oil-dependent polymer sample (HAP). The structure of the polymer molecule is shown in Figure 1.
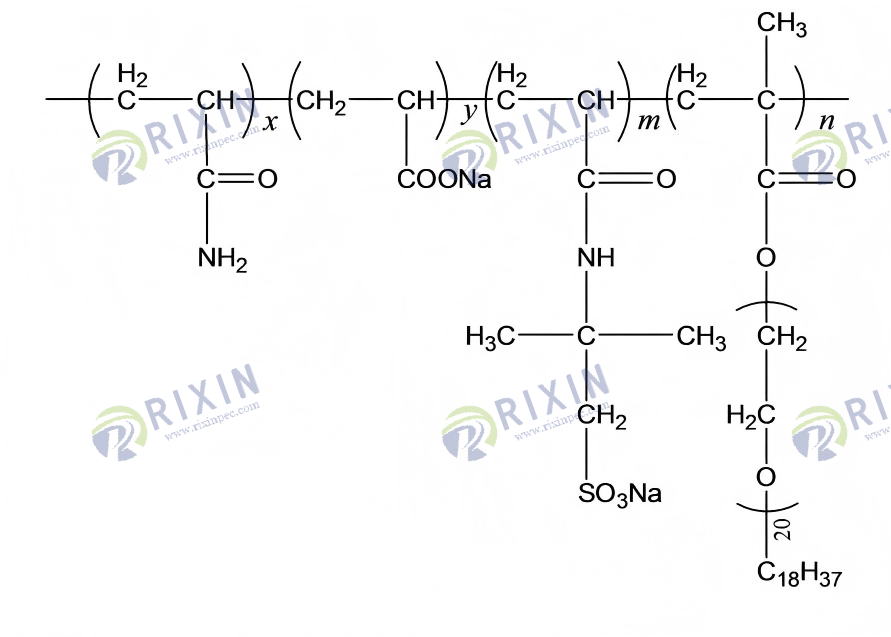
Figure 1. Molecular structure of the amphiphilic polymer HAP
2. Properties of amphiphilic oil-displacing polymers
2. Solubility
The dissolution of polymer macromolecules typically involves two processes: first, water molecules enter the interior of the polymer powder particles, forming a water-absorbing and swellable mass; then, the polymer macromolecules diffuse into the water, with the macromolecular chains in an extended state, giving the solution a certain viscosity. Due to the salt-sensitivity effect of polymers, at high salinity, positively charged metal ions counteract the repulsive effect of —COO- in the polymer, compressing the hydration layer and causing the macromolecular chains to coil, resulting in a significant decrease in viscosity. As shown in Figure 7, at 45℃, with the extension of dissolution time, the apparent viscosity of the 0.175% amphiphilic functional polymer HAP solution shows a rapid increase in the initial stage, followed by a gradual plateau, and stabilizes after 30 min, indicating that HAP has good solubility and high apparent viscosity in seawater. This is because, on the one hand, the —SO3- in the introduced AMPS monomer molecule plays a good anti-salt role; on the other hand, the polyoxyethylene ether nonionic group in the introduced amphiphilic monomer POM also contributes to the rapid dissolution of the polymer.
Figure 7. Change in apparent viscosity of HAP solution with dissolution time.
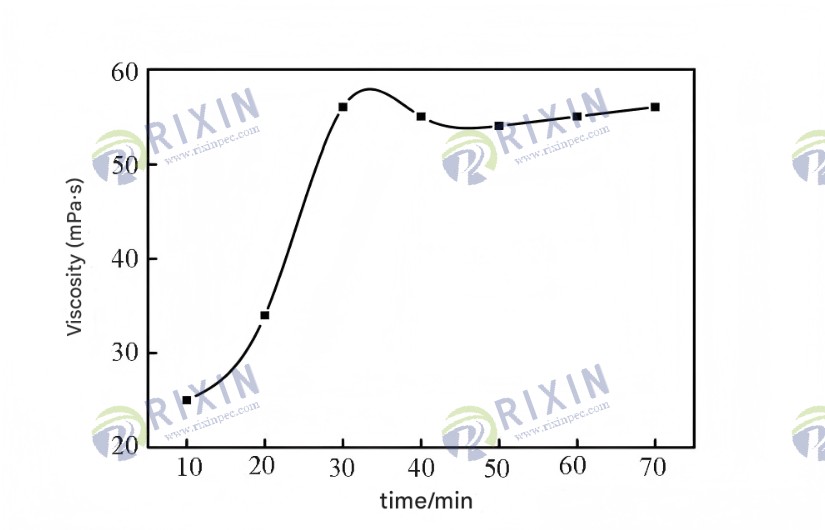
Viscosity is one of the key indicators of polymers used in oil displacement. The stronger the thickening ability of the polymer solution, the lower its concentration can be, resulting in a significant reduction in reagent costs. For comparison, Figure 8 shows the apparent viscosity as a function of concentration for both the amphiphilic oil displacement polymer HAP and a commercially available temperature- and salt-resistant polyacrylamide solution. As shown in Figure 8, the apparent viscosity of both solutions increases with increasing polymer concentration. The viscosity of the temperature- and salt-resistant polyacrylamide solution increases almost linearly with the concentration, while the apparent viscosity of the HAP solution increases slowly at low concentrations. However, when the mass fraction increases to a certain value (0.05%), the curve shows a clear inflection point, and the apparent viscosity rises rapidly. At a mass fraction of 0.175%, the apparent viscosity of the HAP solution is 55.8 mPa·s, significantly higher than that of the commercially available temperature- and salt-resistant polyacrylamide at the same concentration (26.3 mPa·s). The outstanding thickening performance of the amphiphilic oil displacement polymer is mainly due to the salt resistance of the AMPS monomer and the thickening properties of POM. Under seawater conditions, the -SO3- in AMPS effectively inhibits the compression of macromolecular chains by metal ions, while the hydrophobic alkyl long chains in POM exert a good thickening effect.
Figure 8. Viscosity of HAP and commercially available heat-resistant and salt-resistant polyacrylamide solutions as a function of concentration.
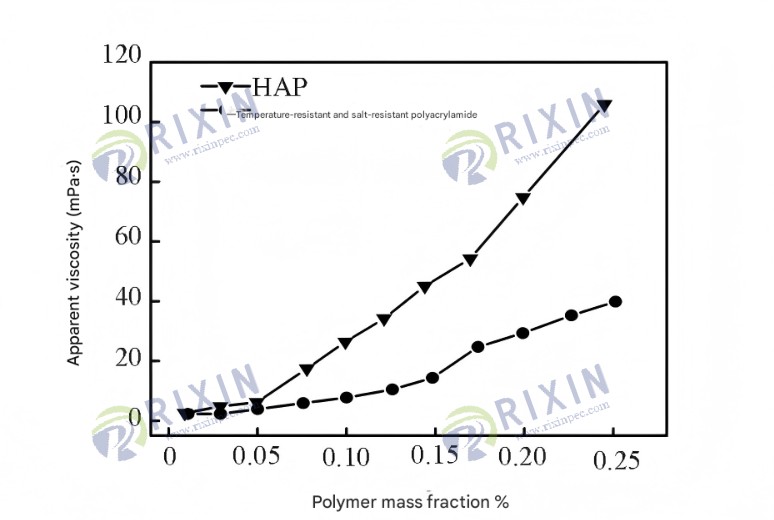
2.2 Salt tolerance
The effect of salinity on the viscosity of the 0.175% polymer solution is shown in Figure 10. As salinity increases, the apparent viscosity of the polymer tends to decrease. Compared with commercially available temperature- and salt-resistant polyacrylamide, the viscosity change of the HAP solution can be divided into three stages. When the salinity is less than 2 g/L, the polymer viscosity decreases rapidly; when the salinity is 2–12 g/L, the apparent viscosity of the HAP solution increases from 95 mPa·s to 139 mPa·s, exhibiting a significant salt thickening phenomenon; as the salinity continues to increase, the negative charge on the polymer macromolecules is further shielded, and the salt thickening is gradually offset, ultimately resulting in a continued decrease in viscosity. Compared with commercially available temperature- and salt-resistant polyacrylamide, the amphiphilic oil-displacing polymer has better salt resistance, which is because the nonionic groups in the amphiphilic monomer POM can mitigate the salt-sensitivity effect of the polymer molecular chain.
Figure 1. Effect of mineralization on apparent viscosity of 0.175% HAP and commercially available temperature- and salt-resistant polyacrylamide solutions.
2.3 Long-term stability
As shown in Figure 11, the viscosity of the 0.175% HAP solution after aging at 75℃ for 90 days still exceeds 30 mPa·s, exhibiting better long-term thermal stability than temperature- and salt-resistant polyacrylamide. This is because the hydrophobic association between HAP molecules can compensate for the viscosity loss caused by polymer degradation. In addition, the hydrophobic segments of HAP contain large side groups, which can improve the thermal stability of the polymer molecular chain.
Figure 1.1 Effect of aging time on the apparent viscosity of 0.175% HAP and commercially available temperature- and salt-resistant polyacrylamide solutions
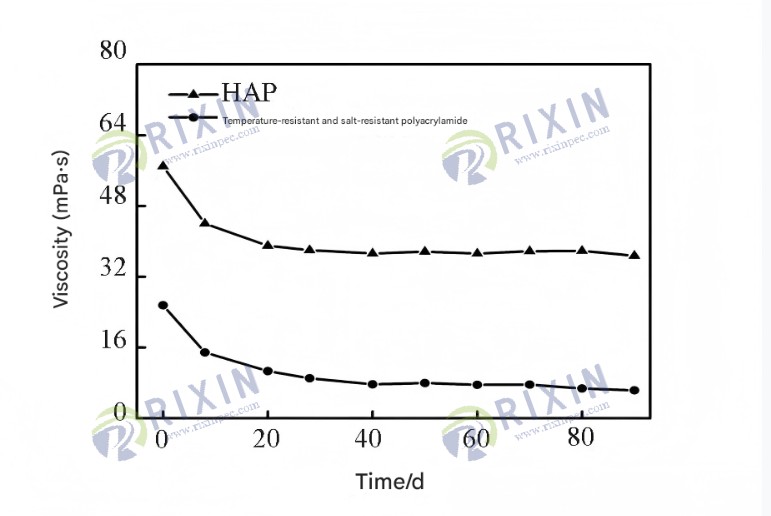
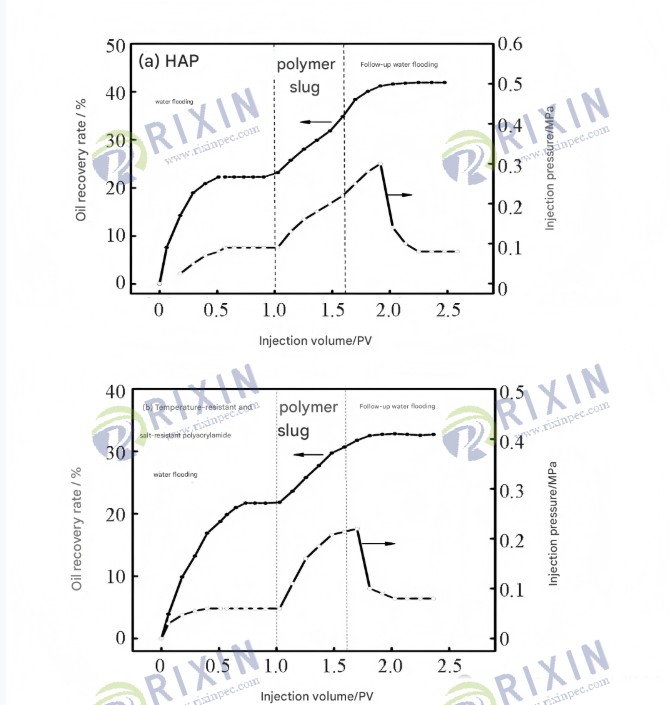
Figure 1.2 Core displacement effects of HAP and commercially available temperature- and salt-resistant polyacrylamide solutions
2.4 Oil displacement performance
Figure 12 shows the changes in oil recovery and injection pressure with injection volume for HAP and commercially available temperature- and salt-resistant polyacrylamide solutions. After injecting 0.6 PV, the highest injection pressure of HAP was 0.31 MPa, while that of commercially available temperature- and salt-resistant polyacrylamide was 0.23 MPa. This is because, at the same concentration, HAP has a higher viscosity and a larger retention capacity at the pore throat than commercially available temperature- and salt-resistant polyacrylamide, thus establishing a higher injection pressure gradient and achieving better sweep efficiency and higher oil recovery. The total oil recovery of cores displaced by 0.175% HAP and commercially available temperature- and salt-resistant polyacrylamide solutions were 42.1% and 33.4%, respectively. Based on water flooding, polymer flooding increased the recovery by 19.7 percentage points and 10.9 percentage points, respectively. The final recovery rate of HAP solution was 8.8 percentage points higher than that of commercially available temperature- and salt-resistant polyacrylamide, demonstrating better oil displacement performance.
3. Conclusion
An amphiphilic oil displacement polymer, HAP, was prepared using acrylamide (AM), acrylic acid (AA), 2-acrylamido-2-methylpropanesulfonic acid (AMPS), and a self-developed long-chain alkyl polyether amphiphilic functional monomer (POM) as raw materials via low-temperature initiation free radical copolymerization. The optimal synthesis conditions were: a total monomer mass fraction of 20%, POM addition of 0.4%–0.8% of the total monomer mass, AMPS addition of 8%, initiator addition of 0.1%–0.4% of the total monomer mass, and an initiation temperature of 0℃. The quaternary copolymer HAP exhibited good solubility, dissolving in simulated Bohai Sea water in 30 min. Its thickening properties, shear resistance, salt resistance, stability, and oil displacement performance at 75℃ were all superior to those of commercially available temperature- and salt-resistant polyacrylamide.