Address
304 North Cardinal St.
Dorchester Center, MA 02124
Work Hours
Monday to Friday: 7AM - 7PM
Weekend: 10AM - 5PM
Address
304 North Cardinal St.
Dorchester Center, MA 02124
Work Hours
Monday to Friday: 7AM - 7PM
Weekend: 10AM - 5PM
The introduction of salt-based ternary peptides into the incoming liquid caused difficulties in handling the new 3-1 solution. While the problem of salt-based ternary peptides and polymer-based peptides was solved by introducing a patent, the dosage was still relatively large. Since most of the water-soluble demulsifier used in the first stage was lost with the drain water, we considered utilizing this portion of the demulsifier and attempting to have it continue to function in the second stage after it had taken effect. To this end, we synthesized LD125 based on the principle of low-temperature demulsification, thus solving the problem of excessive dosage in the second stage.
The new 3-1 processing technology is shown in the figure below (the same for ternary and polymer flooding):
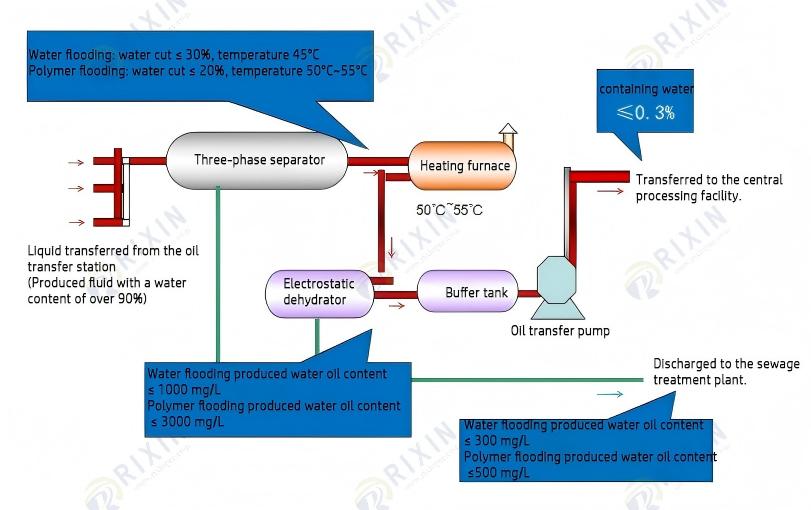
The synthesized LD125 crude oil demulsifier is a water-soluble demulsifier with strong lipophilicity and a low cloud point. When the temperature rises to the polymer’s cloud point, its water solubility relatively decreases while its oil solubility increases, allowing the demulsifier molecules to dissolve in the oil. This utilizes the temperature difference between the first and second stages to enhance the conversion effect. The LD125 crude oil demulsifier is a large molecule with multiple side chains. This molecular structure allows it to lie at the oil-water interface, forming an irregular, relatively loose molecular film. This reduces the strength of the oil-water interfacial film, which is beneficial for improving demulsification ability and speed.
I. Preface
Block polyethers (SP, BP, etc.) using alcohols as initiators include octadecyl alcohol, propylene glycol, glycerol, and pentaerythritol. W/O type crude oil emulsion demulsifiers, such as SP169, use C18H37OH as an initiator. A 1:6:9 (wt) PO-EO-PO polyether is suitable for paraffinic crude oils but not for gum-like, asphaltene-rich, high-heavy, or high-water-content crude oils. AE type (diblock) demulsifiers use polyethylenepolyamines as initiators, while AP type (triblock) demulsifiers are multi-branched, have high wettability and penetration, require low dosage, and offer good demulsification at low temperatures. Demulsifiers using alkylphenol resins as initiators (AF, AR type) such as isobutylphenol, isooctylphenol, nonylphenol, or C9-based mixed alkylphenols have molecular structures similar to natural crude oil emulsifiers and exhibit good penetration and diffusion effects. Demulsifiers reacting block polyethers with polyalkylsiloxanes using polyethylenepolyamines as initiators are less sensitive to the type of crude oil emulsion. Examples include SAE and SAP series, and polymethylphenyl silicone oil-polyoxypropylene polyoxyethylene ether. Ultra-high molecular weight demulsifiers have remarkable demulsifying effects, but must not contain a large number of OH or NH2 groups in the polymer molecule; ideally, each polymer molecule should contain only two or three such groups, and no more than 1/10000. Introducing appropriate hydrophilic or lipophilic groups into high molecular weight polymers, such as polyalkoxyene self-polymers (including self-polymers of mono-epoxides and block copolymers of different epoxys, propylene oxide, butane oxide, and tetrahydrofuran, etc.), and crosslinking them with di-active group compounds, can prepare ultra-high molecular weight polymers. Examples include polyphosphite polyesters and polyamides. Low molecular weight electrolytes and alcohols include: 1) Surfactants: quaternary ammonium salts neutralizing negative charges, dithiocarbamates (producing flocs with Fe2+), and polyamines (adsorbed onto negatively charged interfaces, replacing them); 2) Polymers: diallyl dimethyl ammonium chloride homopolymers, monoallylamine polymers, and other cationic polymers.
O/W type demulsifiers (reverse demulsifiers) are designed for stabilizing the electric double layer. Since most of the active substances and solid particles constituting the emulsion interface film are negatively charged, they must possess strong positive charge to neutralize the negative charge formed by the emulsifier, strong flocculation ability to flocculate emulsion particles, and strong surface activity to reduce the interfacial energy of the emulsion. The chemical demulsification mechanism (phase transfer and reverse phase deformation mechanism) involves the demulsifier contacting the oil-water interface film under the action of thermal and mechanical energy, displacing the natural active substances within the crude oil interface film, and forming a new oil-water interface film; the water-in-oil emulsion can then undergo a reverse phase transformation into an oil-in-water emulsion. The water in the external phase aggregates, and when it reaches a certain volume, it settles out of the oil phase due to the density difference between oil and water.
The main focus of crude oil demulsifier research is on low temperature, rapid processing, and high adaptability. Low temperature aims to save energy; rapid processing improves crude oil handling capacity and shortens processing cycles; high adaptability overcomes the weakness of highly specific demulsifiers, enabling a single demulsifier to meet the needs of different properties of crude oil in different regions. Therefore, researching low-temperature, rapid, and adaptable crude oil demulsifiers is even more important for oilfields entering the high water-cut recovery stage, ensuring crude oil dehydration quality and saving energy. In the early development areas of Daqing Oilfield, the average water content of crude oil has reached over 60%. Due to the continuous increase in total liquid volume, the volume of liquid that each dehydration station needs to handle will far exceed the original design capacity. If the dehydration stations are not rebuilt or expanded, the residence time of water-containing crude oil in the processing equipment must be shortened, and the dehydration speed must be accelerated. Since the specific heat of water is much greater than that of pure oil, if the dehydration temperature is not lowered, energy consumption will inevitably increase daily with the continuous increase in crude oil water content.
Oil-soluble low-temperature demulsifiers suffer from the problem of consuming large amounts of organic solvents. For example, product manuals from companies like Toho Chemical Co., Ltd. in Japan report that Demulfer 334, 424, and Demu-Ifer F-55 are touted as revolutionary low-temperature demulsifiers. However, our experimental research has shown that while these demulsifiers are excellent low-temperature demulsifiers, they require large amounts of lower alcohols such as methanol, ethanol, or isopropanol, or benzene-based organic compounds as solvents to achieve their low-temperature demulsification effect. Currently, the production of these organic solvents in my country is insufficient, hindering their widespread application. Therefore, when developing new low-temperature demulsifiers, we must formulate research plans based on my country’s actual conditions.
The purpose of this experiment was to further study the molecular structure and the relationship between the lipophilic and hydrophilic properties of LD-type demulsifiers and demulsification, and to further develop a low-temperature demulsifier suitable for Daqing Oilfield, namely a water-soluble low-temperature demulsifier. The experiment was conducted in two steps. The first step was the indoor test, which mainly involved the research and screening of indoor formulations. A total of 94 LD series demulsifier formulations were synthesized and evaluated using the indoor crude oil dehydration method. Finally, two better formulations, namely LD125 and LD (128) 236, were selected for detailed comparison. The experiment lasted for six months and obtained multiple data points. During the experiment, various types of demulsifiers from domestic and foreign sources were also compared. The second step was the on-site industrial test of LD125 demulsifier. The experiment was conducted at the Nanwu Joint Station, utilizing the station’s existing equipment and production processes. Comparative tests were performed on three demulsifiers—LD125, LD(129) 236, and SP169—under different temperatures and dosages. The experiment lasted 67 days, during which the station’s dehydration heating furnace was shut down. The lowest dehydration temperature was 38℃, and the lowest dosage was 9.2 PPM. A total of 2880 data points were obtained. The field test showed that LD125 demulsifier performed well in dehydration at a dosage of 10 PPM and a dehydration temperature of 40℃. To further promote the experimental results, an indoor adaptability test of LD125 demulsifier on different crude oils in various oilfields of Daqing was also conducted during this period. This test showed that LD125 demulsifier is suitable for all currently developed oilfields in Daqing, laying the foundation for its future widespread use.
II. The concept of low-temperature dehydration and the molecular structural characteristics of LD125 crude oil demulsifier
Generally, a demulsifier with a demulsification temperature approximately 10°C above the crude oil’s pour point is considered a more advanced low-temperature demulsifier. Besides low temperature, other important factors for evaluating the quality of a demulsifier include high efficiency (reduced dosage), rapid dehydration (fast dehydration), low water content in the treated oil and low oil content in the wastewater, strong adaptability, low price, and ease of use. In developing the LD125 crude oil demulsifier, we comprehensively considered all of the above indicators. From a molecular structure perspective, this demulsifier should possess the following characteristics:
1. LD125 crude oil demulsifier is a large molecule with multiple side chains. This type of molecule can lie at the oil-water interface, forming an irregular, relatively loose molecular film, reducing the strength of the oil-water interfacial film and thus improving demulsification ability and speed. Therefore, our research on LD-type demulsifiers utilizes compounds with multiple active hydrogen atoms as initiators to polymerize epoxides.
2. The molecular structure of LD125 crude oil demulsifier contains multiple hydrophilic groups. This structure of demulsifier has a strong affinity for binding water, which is conducive to the aggregation of small water droplets into larger droplets and their sedimentation.
3. LD125 crude oil demulsifier has a low cloud point. LD-type demulsifiers are nonionic surfactants. The reason these demulsifiers have a low cloud point is that the oxygen atoms on the ether bonds in the polyoxyethylene segments can form hydrogen bonds with water. The formation of these hydrogen bonds is conditional; they form at a certain temperature. When the temperature rises to the polymer’s cloud point, the hydrogen bonds break, water solubility relatively decreases, oil solubility increases, and oil phase concentration increases. To facilitate the use of demulsifiers at low temperatures, based on the above principle, the cloud point of the demulsifier must be lowered to allow the demulsifier molecules to dissolve in the oil at low temperatures (above 40℃). This prevents a large amount of demulsifier molecules from entering the water and being carried away by the discharged wastewater during the first stage of thermochemical dehydration. If a large amount of demulsifier molecules dissolve in the oil, they will enter the electrochemical dehydrator and fully exert their demulsifying effect in the second stage of electrochemical dehydration.
4. LD125 crude oil demulsifier is a water-soluble demulsifier with strong lipophilic properties. Enhanced lipophilicity facilitates the dispersion of demulsifier molecules in water-in-oil emulsions, allowing them to move more quickly to the oil-water interface and exert their demulsifying effect at lower temperatures. In foreign research on low-temperature demulsifiers, most are formulated as oil-soluble, primarily considering their ease of dispersion in water-in-oil emulsions and their ability to quickly reach the oil-water interface for effective demulsification. Therefore, in developing low-temperature demulsifiers, we strive to synthesize demulsifiers with strong lipophilic properties. This is achieved by adjusting the ethylene oxide content in the polymer to appropriately enhance the lipophilicity of the demulsifier while controlling it within the water-soluble range, thus finding the optimal ratio. This is mainly due to its ease of use in oil fields, minimal consumption of organic solvents, and suitability for industrialization.
III. Indoor Tests
We conducted indoor experiments on LD125 demulsifier using two methods. The experimental procedure was as follows: first, a series of LD-type crude oil demulsifiers were synthesized according to different ratios; then, indoor demulsification tests were conducted to screen them and find the optimal formulation. Due to the large number of influencing factors, the data was difficult to reproduce, but the deviation was not large, and the regularity was very obvious.
1. The experimental methods adopted were “orthogonal experimental design” and “function data analysis”.
The orthogonal array used in the “orthogonal experimental method” is “L933”. Based on several years of experience, we divided the structure of LD-type demulsifiers into three factors: lipophilic head (initiator, polypropylene oxide), hydrophilic end (ethylene oxide), and lipophilic tail (polypropylene oxide). The three levels were also determined based on our experience in demulsifier research as: 1, 2, 3; 1, 3, 5; 3, 7, 9. After nine experiments, the data were processed, and the theoretical optimal point was calculated. In total, ten experiments were conducted, resulting in ten different formulations. The evaluation of the formulations’ quality was performed using the indoor crude oil dehydration test method.
The principle of the “function data analysis method” is to represent the experimental points using functional relationships. In a Cartesian coordinate system, the experiment is plotted as a multi-peak curve. The points where the derivative approaches zero are calculated; these points are the extreme values of the multi-peak curve and also the best points in the experiment. This experiment included 37 experimental points and 5 calculation points, varying four factors in the LD-type structure (initiator, propylene oxide, ethylene oxide, and propylene oxide), with each factor varying at 5 levels, i.e., Hn=54.
2. Test instruments, reagents and operating conditions
The polymerization equipment for the demulsifier was a 0.1 cubic meter high-pressure reactor (product of Tianjin No. 2 General Machinery Factory). The polymerization process conditions were: temperature 120-130℃, pressure 3-4 kg/cm², and electromagnetic stirring speed of the polymerization reactor 100 times/min. The reaction rate was 10 g/h for propylene oxide and 12-14 g/h for ethylene oxide. The raw materials for synthesizing the demulsifier were: propylene oxide and ethylene oxide, both industrial grade from Daqing Lamadian Chemical Plant (purity above 98%, aldehyde content <0.1%, water content <0.1%); the initiator was industrial grade from Shanghai Dyeing and Chemical Plant No. 14; and the catalyst, potassium hydroxide, was analytical grade. For details on the indoor demulsification test methods and instruments used, please refer to “Analytical Methods for Nonionic Surfactants”. The dosage of chemicals used in the indoor demulsification tests listed in this article was 100 ppm.
3. Data Analysis and Discussion
1) The 10 data points of the “orthogonal experimental method” are shown in Table 1.
Table 1
| No. | Model | Indoor test demulsification effect | Ethylene oxide % | ||||
| crude oil sample | Temperature (°C) | water color | Purified oil water content % | ||||
| 1 | LD 125 | North Third Station | 50 | cream | 2.15 | 2.75 | 20 |
| 2 | LD (129) 137 | “ | “ | “ | 10.00 | 37.00 | 27 |
| 3 | 159 | “ | “ | clear | 52.00 | 56.00 | 30 |
| 4 | 219 | “ | “ | light yellow | 2.00 | 1,90 | 8 |
| 5 | 233 | “ | “ | cream | 42.00 | 9.00 | 38 |
| 6 | 257 | “ | “ | clear | 3.10 | 22.50 | 36 |
| 7 | 317 | “ | “ | cream | 2.15 | 2.45 | 9 |
| 8 | 339 | “ | “ | “ | 2.15 | 2.75 | 20 |
| 9 | 353 | “ | “ | “ | 40.00 | 25.00 | 45 |
| 10 | 319 | “ | “ | Dark yellow | 2.80 | 2.30 | 7 |
The orthogonal experimental design was conducted with a fixed oleophilic head ratio of 129. Table 1 shows that the ethylene oxide content ranged from 7% to 45% (by weight). The optimal test points in the table are: LD125, LD(129)219, and LD(129)317. Ethylene oxide constitutes 8% to 20% of their compositions. To test these three formulations, LD125, LD(129)219, and Japanese Demulfer 424 crude oil from the West Fourth Station were compared. The test data are shown in Table 2.
Table 2
| No. | Model | Indoor test demulsification effect | Remark | ||||
| crude oil sample | Temperature (°C) | water color | Purified oil water content % | ||||
| 1 | LD 125 | North Third Station | 50 | cream | 3.45 | 3.25 | Produced by our company |
| 2 | LD (129)219 | “ | “ | “ | 3.15 | 3.90 | “ |
| 3 | Demulfer424 | “ | “ | Dark yellow | 2.50 | 2.50 | Japanese East Coast Products |
Table 2 shows that LD125 and LD(129) 219 have similar purified oil quality compared to the oligomer-type demulsifier Demulfer424. Although Demulfer424 is slightly better, the purified oil content is 2.5% for both, but the wastewater quality is poor, and the water color is dark yellow.
(2) Advantages of the “Function Data Analysis Method” experiment
The dehydration data of the 42 test points in the “Function Data Analysis Method” are attached to the summary table (Table 3), which lists the good points that occurred.
Table 3
| No. | Model | Indoor test demulsification effect | Ethylene oxide % | ||||
| crude oil sample | Temperature (°C) | water color | Purified oil water content % | ||||
| 1 | LD (105) 436 | North Third Station | 50 | cream | 2.15 | 2.55 | twenty three |
| 2 | 345 | “ | “ | “ | 2,10 | 2,00 | 33 |
| 3 | LD (169) 112 | “ | “ | “ | 2.85 | 3.14 | 25 |
| 4 | 126 | “ | “ | clear | 2.50 | 2.50 | twenty two |
The percentage of ethylene oxide in the four formulations in Table 3 is basically consistent with the LD125 of the three formulations obtained by the “orthogonal experimental method,” and slightly higher than LD(129) 219 and LD(129) 317. However, it is much lower than the currently used LD(229) 134 and LD(229) 221. This shows that, as a low-temperature demulsifier, the ethylene oxide content should be appropriately reduced.
(3) Relationship between changes in ethylene oxide content and dehydration temperature
Taking one point from the “orthogonal experimental method”, with the head-to-tail ratio of LD125 fixed at 1:3, and varying the ethylene oxide content to 10%, 20%, 30%, 10%, and 50%, indoor demulsification tests were conducted at 60℃, 45℃, and 50℃ respectively. The test results can be plotted as curves (see Figure 1).
Figure 1. Curves from the orthogonal experimental method
The curves show that when the dehydration temperature is 60℃ and the ethylene oxide content is between 10% and 40%, the water content of the purified oil is below 5%. When the dehydration temperature is 50℃ and the ethylene oxide content is between 10% and 30%, the water content of the purified oil is below 8%. When the dehydration temperature is 45℃ and the ethylene oxide content is between 10% and 20%, the water content of the purified oil is below 5%. This fully demonstrates that the dehydration temperature can be correspondingly lowered in LD-type crude oil demulsifiers due to the reduction in ethylene oxide content and the enhancement of lipophilic properties.
Previous experiments have consistently shown that these demulsifiers only have demulsifying effectiveness at temperatures above 55°C. For crude oil from the Beisan Station, demulsification is not possible at temperatures as low as 45°C, and only some oil-soluble demulsifiers can achieve this. However, the above experiments found that both LD125 and LD (129) 236 demulsifiers showed the same dehydration effect as oil-soluble demulsifiers.
The effectiveness of these demulsifiers at low temperatures is primarily due to their enhanced lipophilic properties. For any emulsified crude oil, the dehydration efficiency of any surfactant is related to its interfacial activity. If a surfactant molecule can move quickly to the oil-water interface and form an unstable arrangement there, then the dehydration effect of this surfactant will be ideal. However, the diffusion rate of a surfactant in emulsified crude oil is not solely determined by its molecular structure. The properties of the crude oil, especially paraffin wax and gums, can hinder its diffusion, particularly as the temperature of the emulsified crude oil decreases, significantly reducing the mobility of surfactant molecules. For ultra-high molecular weight oil-soluble demulsifiers, the high chain mobility at low temperatures, coupled with their strong flocculation effect on the dispersed phase, results in effective low-temperature dehydration. Although LD125 and LD236 demulsifiers in Figure 1 are water-soluble, have low molecular weights, and poor flocculation, they have low cloud points, both below 40℃ (below the dehydration temperature). When these are prepared as aqueous solutions and added to emulsified crude oil, they dissolve in the oil due to the breaking of hydrogen bonds and move towards the interface through the oil phase, thereby increasing the dispersion rate and enhancing the demulsification performance. This phenomenon observed in the experiment is consistent with experimental and theoretical research, suggesting that the molecular arrangement of LD125 at the interface at a demulsification temperature of 45℃ may be the desired arrangement of molecules lying on the interface in demulsifier research.
(4) The effect of the ratio of oleophilic head to emulsion breaking effect
In the experiment using the “function data analysis method”, a set of data appeared as shown in Table 4.
Table 4
| No. | Model | Indoor test demulsification effect | ||||
| crude oil sample | Temperature (°C) | water color | Purified oil water content % | |||
| 1 | LD (510)112 | North Third Station | clear | 50 | 7.00 | 2.50 |
| 2 | LD (335)112 | “ | light yellow | “ | 3.00 | 3.00 |
| 3 | LD (170)112 | “ | cream | “ | 2.85 | 3.40 |
| 4 | LD (105)112 | “ | “ | “ | 40.00 | 36.00 |
Table 4 shows that when the lipophilic head ratio varies within the range of 170-300, the water content of the purified oil does not differ significantly, and the water color ranges from clear to light yellow. Although the lowest value of 105 indicates poor dehydration, years of research, production, and application of crude oil demulsifiers have taught us that the lipophilic head ratio of demulsifiers should not be too high. This is because for the stepwise addition polymerization of anionic epoxides, the presence of trace impurities significantly affects the conversion rate of monomers in chain growth. Under the current monomer purity and with potassium hydroxide (KOH) as a catalyst, even if the head ratio is increased, the molecular weight of the polymer will not increase significantly. Furthermore, in factory production, a higher head ratio leads to more intermediates being removed from the reactor, which is detrimental to production continuity and increases byproducts. For example, the presence of small amounts of water and aldehydes in the following initiation growth processes can increase low-molecular-weight byproducts. When the initiator dehydrates under alkaline conditions to form potassium alkoxide, if this water is not completely removed during production, polypropylene glycol may be formed. When aldehydes are present, the following side reactions occur:
CH.CHO+HCH, CHOCH,CH-CH, CHOCH3CH=CHCHO Heating-H₂O OH
CHCHO heating → produces a brownish resinous product.
The H2O removed during the above reaction can also react with propylene oxide to produce polypropylene glycol. As a crude oil demulsifier, polypropylene glycol is inefficient; the resulting resinous product can surround the catalyst, deactivating it or even preventing the reaction from proceeding. To ensure the smooth progress of the reaction, improving the purity of the raw materials is one aspect. Furthermore, from an operational perspective, the intermediate should not be repeatedly removed from the reactor. This not only shortens the production cycle but also ensures product quality. Based on multiple experiments, a 1:129 ratio of lipophilic head to LD-type demulsifier is considered appropriate, and this ratio has been largely adopted in subsequent experiments.
(5) The effect of changes in the oleophilic tail on water color
The adjustment of the lipophilic tail of the LD-type demulsifier was not conducted separately in this experiment. Data from the two experimental methods show that at demulsification temperatures of 45℃, 50℃, and 60℃, the demulsifier structure still follows the previously summarized pattern of a large tail and clear water. However, at dehydration temperatures below 50℃, the tail is too large and the head is too small, resulting in poor water separation, as shown in a set of data in Table 5.
| No. | Model | Indoor test demulsification effect | Ethylene oxide % | ||||
| Crude oil sample | Temperature (°C) | Water color | Purified oil water content % | ||||
| 1 | L D ( 189 ) 116 | North Third Station | 50 | clear | 29.50 | 39.50 | 10 |
| 2 | 136 | “ | “ | clear | No water comes out | 30 | |
| 3 | 156 | “ | “ | clear | 47.00 | 49.00 | 40 |
| 4 | 176 | “ | “ | cream | No water comes out | 50 | |
Table 5 shows that the effect is not ideal, but it also reflects that the small head and excessively large tail result in excessive water content in the purified oil. If the head-to-tail ratio is properly selected, the effect of clean oil and clear water can also be achieved (see Table 6).
| No. | Model | Indoor test demulsification effect | Ethylene oxide % | ||||
| Crude oil sample | Temperature (°C) | Water color | Purified oil water content % | ||||
| 1 | LD (₁₂₉)236 | North Third Station | 50 | clear | 7.10 | 7.15 | 30 |
| 2 | LD (₁₇₂)126 | “ | “ | clear | 2.50 | 2.50 | twenty two |
| 3 | LD (₁₈₀)359 | “ | “ | clear | 4.50 | 4.60 | 30 |
(6) Solvent addition test
This experiment was conducted after the basic adjustment of the LD series demulsifiers was completed. The four formulations in Table 7 were considered to have good dehydration effects at 50℃. The solvent was methanol (CH₃OH), accounting for 33% of the total volume, with 2% H₂O added. The demulsification dosage was 65%, and the preparation method was the same as the existing demulsifier solvent addition method.
Table 7
| No. | Model | Indoor test demulsification effect | Remark | ||||
| Crude oil sample | Temperature (°C) | Water color | Purified oil water content % | ||||
| 1 | LD 125 | North Third Station | 50 | light yellow | 7.00 | 12.00 | Average of several samples |
| 2 | LD (₁₂₉)236 | “ | “ | clear | 14.00 | 15.50 | “ |
| 3 | LD (₁₄₀)436 | “ | “ | light yellow | 14.40 | 22.00 | “ |
| 4 | LD (₁₈₀)345 | “ | “ | clear | 26.40 | 24.40 | “ |
Table 7 shows that the dehydration effect of this type of demulsifier significantly decreased after adding solvent, and the water color darkened and the effluent flow rate slowed down, which is the same as that of LD(₂₂₆)221 demulsifier. To verify the effect of dosage changes on dehydration, an experiment was conducted where the above-mentioned demulsifier was added to a solvent while maintaining the original dosage of 100 ppm. The experimental results show that as long as the appropriate dosage is maintained, this type of demulsifier still has excellent dehydration performance at low temperature, quickly, cleanly removes oil, and clears water after adding 35% methanol water solvent.
4. Conclusions of Indoor Tests
Through multiple synthesis and screening experiments, we believe that LD125 crude oil demulsifier is a good low-temperature demulsifier for dehydrating Daqing crude oil, and its dehydration effect is also very good at higher temperatures. Tables 8-13 list a large amount of experimental data.
Summary of LD125 Crude Oil Demulsifier Dehydration Tests
Table 8
| No. | Analysis date ( year, month, day ) | Sample number | Dehydration temperature (°C) | Purified oil water content (%) | Water color | Sample source | Crude oil sample source | Remark | |
| 1 | 17. 9.22 | 17 107 | 60 | 1.75 | 1.50 | cream | Our company | North Third Station | Fast water output |
| 2 | 17. 9.25 | “ | 60 | 3.20 | 2.10 | clear | “ | “ | |
| 3 | 17 , 10.5 | “ | 50 | 2.15 | 2.75 | light yellow | “ | “ | |
| 4 | 17 , 10.10 | “ | 50 | 2.90 | 2.10 | cream | “ | “ | |
| 5 | 17 , 10, 11 | “ | 50 | 3.45 | 3.2 5 | “ | “ | Xisi Station | |
| 6 | 17 , 11, 30 | “ | 50 | 1.20 | 1.20 | clear | “ | La43 * Station | |
| 7 | 18 , 1.17 | 18 116 | 50 | 2.40 | 2.55 | cream | “ | North Third Station | |
| 8 | 18 , 2.14 | “ | 60 | 2.50 | 2.25 | “ | “ | “ | |
| 9 | 18.2.15 | “ | 50 | 2.40 | 2.60 | “ | “ | “ | |
| 10 | 18 , 2.15 | “ | 45 | 4.50 | 2.30 | “ | “ | “ | |
| 11 | 18 , 2, 17 | “ | 45 | 2.50 | 4.50 | clear | “ | “ | |
| 12 | 18.3.1 | “ | 45 | 3.55 | 2.85 | clear | “ | “ | |
| 13 | 18.3.2 | “ | 50 | 3.00 | 6.00 | light yellow | “ | “ | |
| 14 | 18.3 , 3 | “ | 50 | 1.95 | 3.75 | cream | “ | “ | |
| 15 | 18 , 3, 9 | “ | 45 | 2.05 | 2.05 | clear | La33 * Station | “ | |
| 16 | 18 , 3, 7 | “ | 45 | 2.85 | 3.55 | cream | Our company | La33 * Station | |
| 17 | 18 , 3.22 | “ | 45 | 2.00 | 2.00 | clear | “ | West Oil Depot | |
| 18 | 18 , 3, 28 | “ | 50 | 2.70 | 3.30 | Slightly yellow | “ | North Third Station | Aged oil |
| 19 | 18 , 4, 19 | “ | 60 | 2.50 | 2.15 | “ | “ | “ | |
| 20 | 18 , 4, 25 | “ | 60 | 1.85 | 1.70 | “ | “ | “ | |
| twenty one | 18 , 5, 9 | 18 53 | 47 | 2.00 | 2.60 | “ | “ | “ | |
| twenty two | 18 , 5.9 | 18 52 | 47 | 4.50 | 4.25 | “ | “ | “ | |
| twenty three | 18 , 5, 10 | 18 51 | 47 | 2.80 | light yellow | “ | “ | ||
| twenty four | “ | 18 214 | “ | 4.00 | “ | “ | “ | ||
| 25 | “ | 18 112 | “ | 3.00 | “ | “ | “ | ||
| 26 | “ | 18 53 | “ | 2.50 | “ | “ | “ | ||
| 27 | “ | 18 215 | “ | 2.90 | “ | “ | “ | ||
| 28 | “ | 18 52 | “ | 2.10 | “ | “ | “ | ||
| 29 | 18 , 6, 16 | 37* | 60 | 4.50 | 3.75 | cream | Dalian | La33 * Station | |
| 30 | 18.6.19 | twenty three* | 45 | 4.40 | 5.50 | clear | “ | La33 * Station | transition zone oil |
| 31 | “ | 18* | “ | 5.50 | 5.75 | cream | “ | “ | |
| 32 | 18 , 6.20 | 13* | “ | 3.75 | 5.40 | “ | “ | “ | |
| 33 | 18 , 7.7 | 1# | “ | 3.50 | 3.00 | “ | lahua | North Third Station | |
| 34 | “ | 2# | “ | 3.40 | 2.85 | light yellow | “ | “ | |
| 35 | 18 , 7.8 | 14* | “ | 3.70 | 3.50 | “ | “ | “ | |
| 36 | “ | 6# | “ | 4.00 | 4.50 | “ | “ | “ | |
| 37 | 18 , 7.21 | 37* | “ | 4.00 | 4.00 | clear | Dalian | La33 * Station | |
| 38 | 18 , 7.27 | 10* | “ | 5.00 | 4.50 | clear | “ | “ | |
| 39 | “ | 2# | “ | 4.50 | 5.50 | clear | “ | “ | |
| 40 | 18 , 7, 31 | 13* | “ | 2.60 | 2.10 | cream | “ | Nanwulian | |
| 41 | 18 , 8.2 | 13# | “ | 3.00 | 2.00 | “ | “ | “ | |
| 42 | 78, 8.3 | 13# | “ | 2.40 | 1.80 | clear | “ | “ | |
Table 9
| No. | Model | Indoor test demulsification effect | Ethylene oxide % | ||
| Water color | Purified oil water content % | ||||
| 1 | LD (129)219 | light yellow | 2.70 | 5.75 | 7 |
| 2 | 216 | cream | 1.70 | 1.90 | 10 |
| 3 | 136 | “ | 3.00 | 3.20 | 30 |
| 4 | LD (109) 436 | “ | 1,20 | 1,30 | twenty three |
| 5 | LD 125 | 2.50 | 2,25 | 20 | |
| 6 | LD (1)113 | clear | 1.40 | 1.50 | 25 |
| 7 | LD (109)345 | cream | 1.40 | 1,35 | 20 |
| 8 | LD (1)236 | “ | 3,50 | 2.25 | 30 |
| 9 | 149 | clear | A small amount of water was discharged. | 30 | |
| 10 | 256 | clear | 1.80 | 1.85 | 40 |
| 11 | 156 | clear | 1.85 | 1.95 | 40 |
| 12 | 169 | cream | 4.20 | 4.30 | 40 |
| 13 | 286 | clear | 48.00 | 11.50 | 50 |
Table 9 shows the data for a dehydration temperature of 60℃ and a settling time of 1.5 hours.
Table 10
| No. | Model | Indoor test demulsification effect | Ethylene oxide % | ||||
| Temperature (°C) | Time | Water color | Purified oil water content % | ||||
| 1 | LD (123)216 | 50 | 1,5 | light yellow | 2.12 | 2,16 | 10 |
| 2 | LD 125 | “ | “ | cream | 2,40 | 2,60 | 20 |
| 3 | LD (1)236 | “ | “ | clear | 7,10 | 7.15 | 30 |
| 4 | 256 | “ | “ | Unable to dehydrate | 40 | ||
| 5 | 286 | “ | “ | clear | 54.00 | 49.00 | 50 |
| 6 | 116 | “ | “ | clear | 29.50 | 39.00 | 10 |
| 7 | 126 | “ | “ | cream | 2.70 | 2.20 | 20 |
| 8 | 136 | “ | “ | clear | A small amount of water was discharged. | 30 | |
| 9 | 156 | “ | “ | clear | 47.00 | 49.00 | 40 |
| 10 | 176 | “ | “ | milky yellow | A small amount of water was discharged. | 50 | |
| 11 | 119 | “ | “ | cream | “ | 10 | |
| 12 | 139 | “ | “ | clear | “ | 20 | |
| 13 | 149 | “ | “ | clear | “ | 30 | |
| 14 | 169 | “ | “ | clear | “ | 40 | |
| 15 | 199 | “ | “ | clear | “ | 50 | |
Table 11
| No. | Model | Indoor test demulsification effect | Ethylene oxide % | ||||
| Temperature (°C) | Time h | Water color | Purified oil water content % | ||||
| 1 | LD (₁₂₉)219 | 45 | 1.5 | light yellow | 4.05 | 3.00 | 7 |
| 2 | LD 125 | “ | “ | cream | 3.55 | 2.85 | 20 |
| 3 | LD (₁₂₉)236 | “ | “ | clear | 3.45 | 3.30 | 30 |
| 4 | 256 | “ | “ | clear | 44.00 | 40 | |
| 5 | 286 | “ | “ | clear | 48.00 | 50 | |
| 6 | 126 | “ | “ | clear | 18.00 | 42.00 | 20 |
| 7 | 317 | “ | “ | cream | 33.00 | 29.00 | 9 |
| 8 | 136 | “ | “ | No water comes out | 30 | ||
Table 12
Summary table of data from the “orthogonal experimental method”
| No. | Model | Indoor test demulsification effect | ||||
| Crude oil sample | Temperature (°C) | Water color | Purified oil water content % | |||
| 1 | LD (₁₂₉)137 | 60 | 1.5 | clear | 4.35 | 21.00 |
| 2 | LD 125 | “ | “ | cream | 1.75 | 1.50 |
| 3 | LD (₁₂₉)159 | “ | “ | clear | 2.00 | 2.85 |
| 4 | 233 | “ | “ | clear | 6.50 | 27.50 |
| 5 | 219 | “ | “ | clear | 2.15 | 2.00 |
| 6 | 257 | “ | “ | clear | 3.55 | 4.75 |
| 7 | 317 | “ | “ | cream | 1.95 | 1.95 |
| 8 | 353 | “ | “ | 1.35 | 1.60 | |
| 9 | LD (₈)137 | “ | “ | clear | 2.40 | 2.40 |
| 10 | LD (₈)319 | “ | “ | cream | 5.20 | 1.85 |
| 11 | LD (₈)159 | “ | “ | clear | 1.10 | 3.90 |
| 12 | 233 | “ | “ | cream | 1.55 | 2.60 |
Table 13
Summary Table of Data on “Function Data Analysis Method”
| No. | Model | Indoor test demulsification effect | Ethylene oxide % | ||||
| Temperature ℃ | Time h | Water color | Purified oil water content % | ||||
| 1 | LD (40)111 | 50 | 1.5 | clear | 29.00 | 37.50 | 33 |
| 2 | LD(105)1:1.6:3.7 | 50 | 1.5 | cream | 3.75 | 3.00 | 25 |
| 3 | LD(17)1:1.5:3 | 50 | 1.5 | cream | 8.50 | 2.25 | 27 |
| 4 | LD(235) 1:1.2:2.3 | 50 | 1.5 | light yellow | 3.00 | 3.00 | 27 |
| 5 | LD(300)1:1:2 | 50 | 1.5 | clear | 7.00 | 2.50 | 25 |
| 6 | LD(170)1:0.24:0,24 | 50 | 1.5 | light yellow | 1.20 | 1.50 | 16 |
| 7 | LD(40)3:1:1 | 50 | 1.5 | Slightly yellow | 8.00 | 42.00 | 25 |
| 8 | LD(40)1:19.25:1 | 50 | 1.5 | light yellow | 50.00 | 50.00 | 91 |
| 9 | LD(40)1:1:38 | 50 | 1.5 | light yellow | 40.00 | 50.00 | 25 |
| 10 | LD(300)1:0.133:0.133 | 50 | 1.5 | Slightly yellow | 29.00 | 38.00 | 10 |
| 11 | LD(40)5:1:1 | 50 | 1.5 | cream | 40.00 | 46.0 | 14 |
| 12 | LD(40)1:37.5:1 | 50 | 1.5 | / | 45.00 | 50.00 | 94 |
| 13 | LD(40)1:1:75 | 50 | 1.5 | light yellow | 26.00 | 50.00 | 13 |
| 14 | LD(170) 1:1.2:2.3 | 50 | 1.5 | cream | 2.85 | 50.00 | 27 |
| 15 | LD(105)1:1.3:2.7 | 50 | 1.5 | cream | 4.40 | 50.00 | 26 |
| 16 | LD(105)1:3,7:3,7 | 50 | 1.5 | clear | 38.00 | 50.00 | 45 |
| 17 | LD(105) 1:1, 9:7.2 | 50 | 1.5 | clear | 27.50 | 50.00 | 18 |
| 18 | LD(235)1:0.65:1.6 | 50 | 1.5 | light yellow | 2.00 | 50.00 | 19 |
| 19 | LD(105)1: 5.4; 3.7 | 50 | 1.5 | clear | 50.00 | 50.00 | 53 |
| 20 | LD(105) 1:1, 9:10.8 | 50 | 1.5 | clear | 38.00 | 50.00 | 13 |
| twenty one | LD(235) 1:1, 1:7.2 | 50 | 1.5 | / | / | / | 26 |
| twenty two | LD(170)1:1.1:2.2 | 50 | 1.5 | clear | 3.00 | 2.75 | 26 |
| twenty three | LD(170)1:22:3 | 50 | 1.5 | clear | 55.00 | 55.00 | 35 |
| twenty four | LD(:70)1:1,5:4,4 | 50 | 1.5 | light yellow | 3.00 | 3.70 | twenty two |
| 25 | LD(300) 1:0, 9:1.7 | 50 | 1.5 | cream | 55.00 | 2.40 | 25 |
| 26 | LD(170) 1:0, 9:1.8 | 50 | 1.5 | light yellow | 3.00 | 2.80 | twenty four |
| 27 | LD(170) 1:2, 9:3, 0 | 50 | 1.5 | clear | 55.00 | 50.00 | twenty two |
| 28 | LD(170) 1:1.5:5.9 | 50 | 1.5 | clear | 3.00 | 2.25 | 18 |
| 29 | LD(105)1:0,38:0,38 | 50 | 1.5 | light yellow | 55.00 | 2.70 | twenty two |
| 30 | LD(40)2:1:1 | 50 | 1.5 | cream | 3.00 | 36.00 | 25 |
| 31 | LD(40)1:10, 12:1 | 50 | 1.5 | light yellow | 55.00 | 48.00 | 33 |
| 32 | LD( (40) 1:1:19, 5 | 50 | 1.5 | Slightly yellow | 3.00 | 42.00 | 5 |
| 33 | LD(300)1:0.675:1,3 | 50 | 1.5 | light yellow | 55.00 | 2.60 | twenty three |
| 34 | LD(105)1:0.77:1,5 | 50 | 1.5 | cream | 3.00 | 1.70 | twenty four |
| 35 | LD(105) 1:7, 14:3.7 | 50 | 1.5 | light yellow | 55.00 | 40.00 | 60 |
| 36 | LD(105) 1:1, 9:14.3 | 50 | 1.5 | light yellow | 3.00 | 1.70 | 11 |
| 37 | LD(140) 3.5:1:1 | 50 | 1.5 | light yellow | 55.00 | 40.00 | 18 |
| 38 | LD(260) 2.6:2:3.9 | 50 | 1.5 | cream | 3.00 | 1.70 | twenty four |
| 39 | LD(105)1:1:2 | 50 | 1.5 | clear | 55.00 | 40.00 | 25 |
| 40 | LD(105)1:1:2 | 50 | 1.5 | clear | 3.00 | 1.70 | 26 |
| 41 | LD(235)2:2:4.3 | 50 | 1.5 | clear | 55.00 | / | twenty four |